A gas at 473Kand 43 atm has a molar volume 28 percent larger than that calculated from the perfect gas law. Calculate the molar volume (dm³mol-4) of the gas. O a 1.16 Ob. 14.08 Oc. 2.06 O d. 0.70
A gas at 473Kand 43 atm has a molar volume 28 percent larger than that calculated from the perfect gas law. Calculate the molar volume (dm³mol-4) of the gas. O a 1.16 Ob. 14.08 Oc. 2.06 O d. 0.70
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 92AP: Unreasonable results. (a) Find the temperature of 0.360 kg of water, modeled as an ideal gas, at a...
Related questions
Question
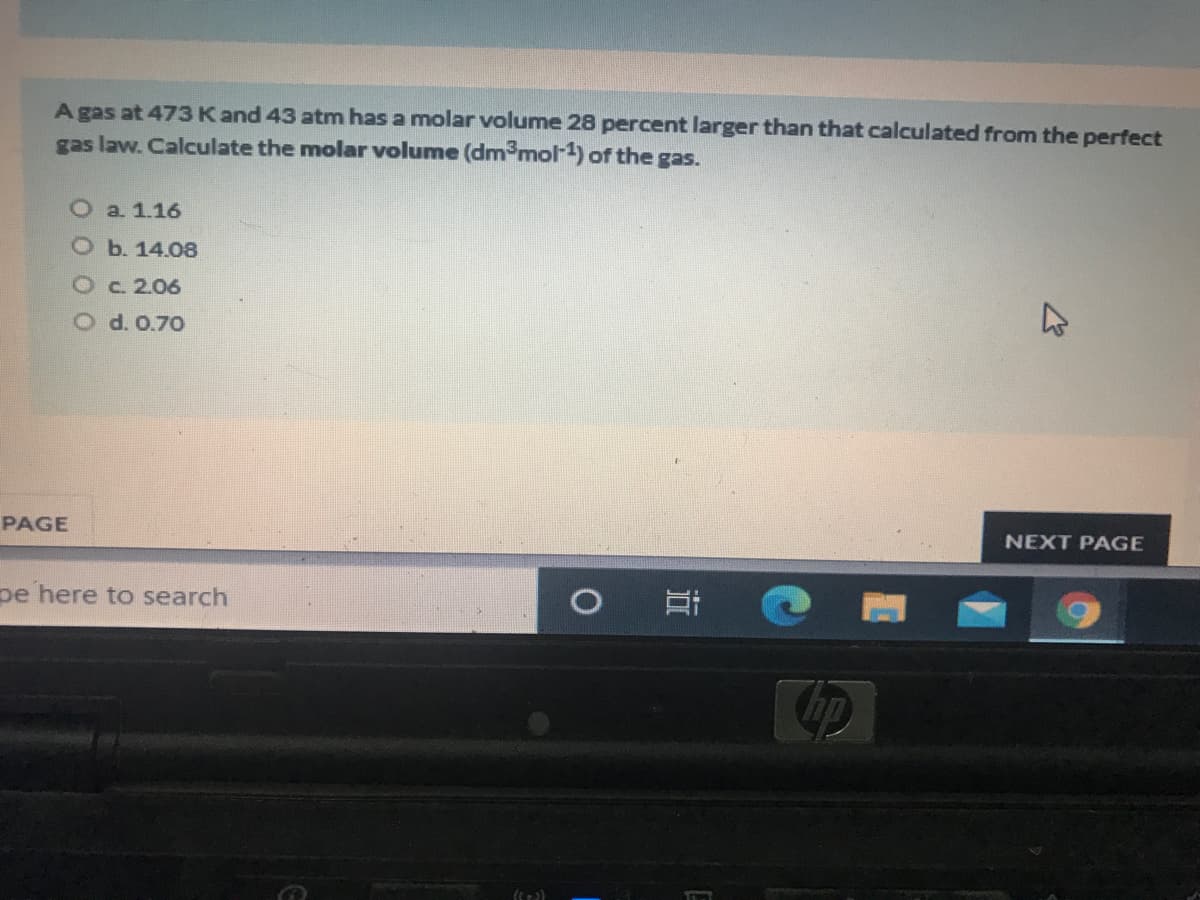
Transcribed Image Text:A gas at 473Kand 43 atm has a molar volume 28 percent larger than that calculated from the perfect
gas law. Calculate the molar volume (dm3mol-1) of the gas.
O a 1.16
O b. 14.08
Oc.2.06
O d. 0.70
PAGE
NEXT PAGE
pe here to search
hp
近
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College