A volume of air (assumed to be an ideal gas) is first ooled without changing its volume and then expanded vithout changing its pressure, as shown by the path abc n the figure (Figure 1). Take the graduation » = 3.0 × 10 Pa and the graduation V = 0.06.
A volume of air (assumed to be an ideal gas) is first ooled without changing its volume and then expanded vithout changing its pressure, as shown by the path abc n the figure (Figure 1). Take the graduation » = 3.0 × 10 Pa and the graduation V = 0.06.
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 57P: An ideal gas expands isothermally along AB and does 700 J of work (see below). (a) How much heat...
Related questions
Question
Transcribed Image Text:Part D
If the air instead expands from state a to state c by the straight-line path shown, how much heat
does it exchange with its surroundings?
Express your answer in joules using two significant figures.
?
Qac = 8000
J
Submit
Previous Answers Request Answer
X Incorrect; Try Again; 2 attempts remaining
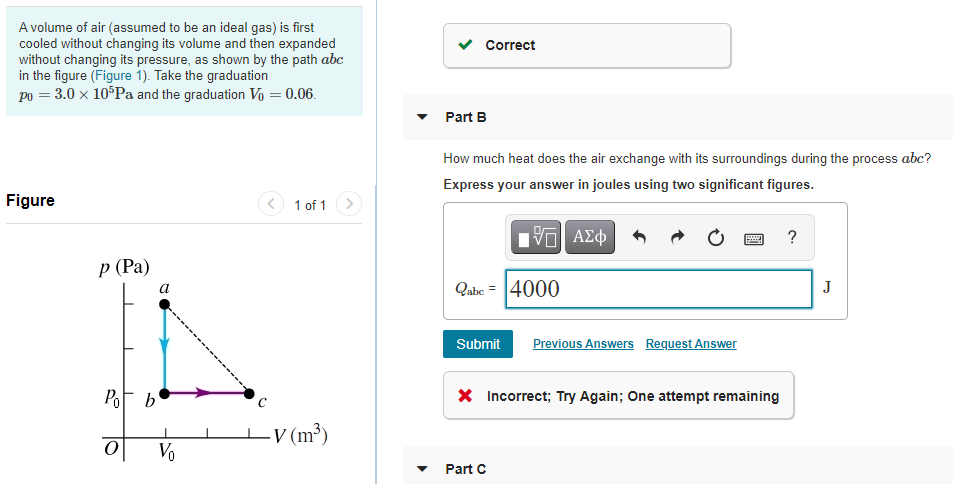
Transcribed Image Text:A volume of air (assumed to be an ideal gas) is first
cooled without changing its volume and then expanded
without changing its pressure, as shown by the path abc
in the figure (Figure 1). Take the graduation
po = 3.0 x 10°Pa and the graduation V = 0.06.
Correct
Part B
How much heat does the air exchange with its surroundings during the process abc?
Express your answer in joules using two significant figures.
Figure
< 1 of 1>
Hνα ΑΣφ
Р (Рa)
Qabe = 4000
J
a
Submit
Previous Answers Request Answer
PoF b'
X Incorrect; Try Again; One attempt remaining
V (m³)
Vo
Part C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning