a) H b) 6 d) 1) CH3MgBr 2) CH CH₂OH 1) Br₂, H₂O 2) pyridine er TYCH CH2MgBr 2) H₂O の ملی 1) (CH3CH2)2CuLi 2) CH,CH₂OH TBDMSO MgBr 1) H 2) H₂O*
Q: of OH Starting materials 1 Br H,C OH 2 Br 6 Reagents a Mg / dry ether b 1. CO2 2. acidic workup c…
A: Find out suitable reagent for this reaction.
Q: Give IUPAC and the common ame for the following amine. Careful with spelling. (CH3CH2CH2)2NCH2CH3 I…
A: The common name and the IUPAC name of the given compound has to be determined.
Q: Devise a multistep synthetic route for the following transformation. You may only use three-carbon…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Design a buffer that has a pH of 10.12 using one of the weak base/conjugate acid systems shown…
A: Given,The pH = 10.12The volume of the buffer = 1.00 LThe molarity of the base = 1.00 M
Q: 9. Select reagents from the table to show how you would carry out these transformations. You should…
A:
Q: Rank by stability of arenium ion (also called sigma complex) upon bromination. The renium ion that…
A:
Q: in the oxidation of cyclohexanol to cyclohexanone, what purpose does the acetic acid serve?
A: Given reaction, Oxidation of cyclohexanol to cyclohexanoneIn the given question, it has been…
Q: Predict the product and draw a detailed mechanism for the transformation.
A: The given reaction is a Dieckmann Condensation reaction and the product is given below. Explanation:
Q: Do the reactions below proceed in good yield from left to right as shown? a) b) c) Na + aqueous HCI…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: We have to draw the curved electron pushing arrows.
Q: Using electrons and/or lone pairs in the following conjugated system, create a resonance form by…
A: When all the properties of a molecule cannot be explained by a single structure, we draw more than…
Q: What is the major product of the following reaction? EIO OEI 1. NaOEt 2. H₂O+ Give detailed…
A: The Dieckmann condensation is a reaction that occurs when diesters react with a base to produce…
Q: Question 24 Identify the Major and ALL Minor product(s) that are expected for each of the following…
A: The objective of the question is to predict the product formed in the following reaction given.The…
Q: Oxygen can be produced by the thermal decomposition of mercuric oxide: heat 2 HgO(s) 2 Hg() + O2(g)…
A: Given,The balanced reaction:
Q: Substance Specific Heat Capacity (J/g °C) 1. ethylene glycol 2.47 2. brass 0.377 3. glass 0.837 4.…
A: Given,specific heat capacity of ethylene glycol = 2.47 J/g.°Cspecific heat capacity of brass = 0.377…
Q: The equilibrium constant, K, for the following reaction is 5.10×10-6 at 548 K. NH4Cl (s)…
A: The objective of the question is to calculate the concentrations of the two gases.
Q: 4.4 (Pages 149-155); Watch KCV 4.4, IWE 4.6. Elemental phosphorus reacts with chlorine gas according…
A: Given reaction,Mass of Molar mass of Mass of Molar mass of Excess reactant left= ?
Q: Provide the enantiomeric Diels-Alder adducts that would form from the following reaction along with…
A:
Q: Draw the mechanism for the following SN2 reactions. Redraw the reactants. a. + -OCH3 H
A:
Q: Question 45.e of 45 A sample of helium is initially at 565 torr in a volume of 2.85 L. At 24.7 °C…
A: The density of He is : 0.122 g/L and Ar is : 1.22 g/L. It is given that both He and Ar has equal…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A:
Q: PyMol commands needed to make the following image of a helix (residues 84-104) found in the Ras…
A: The objective of the question is to generate a specific image of a helix (residues 84-104) found in…
Q: The equilibrium constant, Kc , for the following reaction is 0.0290 at 1150 K. 2SO3= 2SO2 + O2 If…
A: The equilibrium constant, often denoted as Kc, is a quantitative measure of the extent of a…
Q: < H 1) NaOH / H₂O 2) dilute HCI Select to Draw
A: Given that, the reaction is:
Q: Match the Term with the definition 1. Equilibrium position 2. Chemical equilibrium 3. Reaction…
A: The objective of the question is to match the terms with their correct definitions, calculate the…
Q: A new drug has been discovered which inhibits the reaction catalyzed by enzyme A. Based on the graph…
A: Explanation:- ● Competitive inhibition takes place, when the molecules are similar to the substrate…
Q: .Br 袋 NO2 Pd, PPhg, base .NH2
A:
Q: What is the major product and mechanism of the following reaction? www NaSH DMSO
A: In the given reaction alkyl halide reacts with sodium hydrosulfide to form a thioether.
Q: 3. Give the reagents needed to form the indicated products: CN CN
A: In the given example the first step is elimination reaction to form a double bond. In the second…
Q: Consider the combustion of liquid C₅H₈ in oxygen gas to produce carbon dioxide gas and water vapor.…
A: The objective of the question is to find out the quantity in moles of C5H8 that were burned in the…
Q: Check the box under each structure in the table that is an enantiomer of the molecule shown below.…
A: Constitutional isomers: Molecules with the same molecular formula but different…
Q: 9.38 Classify each of the following as exothermic or endothermic, and give the AH for each: a.…
A: Given:
Q: HO OH vs. HO HỌỌC VS. HOOC HOOC pKa's: 2.83; 3.03, 4.54, 5.69 OH .COOH pKa's: 1.92, 3.02, 4.38, 6.23
A: Diaprotic acid: A diprotic acid is an acid that can donate two protons or H+ ions to its aqueous…
Q: Draw the starting structure that would lead to the major products shown under these conditions.
A:
Q: Curved arrows are used to illustrate the flow of electrons. Use the reaction conditions provided and…
A: The given curved arrows and reaction conditions produce the following intermediate benzoate ion as…
Q: The synthesis of nitrous oxide gas (n2o) for anesthetic uses can be done by combining ammonia and…
A: The objective of the question is to determine the amount of oxygen needed to react completely with…
Q: Complete the table below, which lists information about some diatomic molecules or molecular ions.…
A: Bond order = When the bond order is either equal to or greater than 1, then the molecule is said to…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A: An arrow always depicts from a region of high electron density to low electron density ; that is the…
Q: Identify the labeled chiral carbons as R or S in the molecules shown below. HO H C... OH 1) CH3 H 2)…
A: Given,The molecules:
Q: 22. Draw the major product of the following SN2 reaction. Draw a mechanism, showing the transition…
A: The bimolecular nucleophilic substitution reaction is a concerted reaction that occurs in a single…
Q: How to obtain 2-bromo-4-nitro-1-(propan-2-yl)benzene from (propan-2-yl)benzene.
A: GivenReactant: (propan-2-yl)benzene or isopropyl benzene Product :…
Q: Your Current Score = 6 Give IUPAC name for t N CH3 CH3CH2CHCH2CH3 The IUPAC name is n-cyc
A: In the case where there is a cycloalkyl and alkyl both the groups in an amine molecule irrespective…
Q: Calculate the lowest absorption frequency for a linear molecule containing five conjugated double…
A: Given a linear molecule containing five conjugated double bonds.The length of -bond system is 1.05…
Q: multi-step synthesis. Ignore any inorganic byproducts formed. 1. BH 3, THF Br Br a NaNH2 Select to…
A: Product of following reaction can be made by applying appropriate reaction mechanism.
Q: Which of these statements about the mass of subatomic particles is true? Responses The…
A: The question is asking us to identify the correct statement about the mass of subatomic particles.…
Q: Molecule A C1819 NOS pKa = = 9.3 Which of these pairs of species is more abundant in a solution of…
A: Answer:Any specie that gains H+ ion in its solution is called Bronsted-Lowry base and the specie…
Q: Macmillan Learning Complete the reaction. Add hydrogen atoms and charges to the appropriate atoms.…
A: The compounds having -CONR2 group where R can be alkyl groups or H atoms are identified as amides.…
Q: Initiation Propagation Termination Br2 hv Initiation NBS hv Br HBr H2O2 Initiation Propagation…
A: The objective of the question is to find the initiation, propagation, and termination steps of the…
Q: • Calculate the molar solubility of Ag3PO4 if the Ksp = 1.8 x 10-18 10-5 MAGNO solution are mixed…
A: Since you have asked multiple question, we will solve the first question for you.If you want any…
Q: Indicate the mechanism of Hofmann degradation of the compound N-(2-phenylethyl)butan-1-amine.…
A: The objective of the question is to understand the mechanism of Hofmann degradation of the compound…
Give the major product of the following reaction for letter c) and explain the mechanism/process
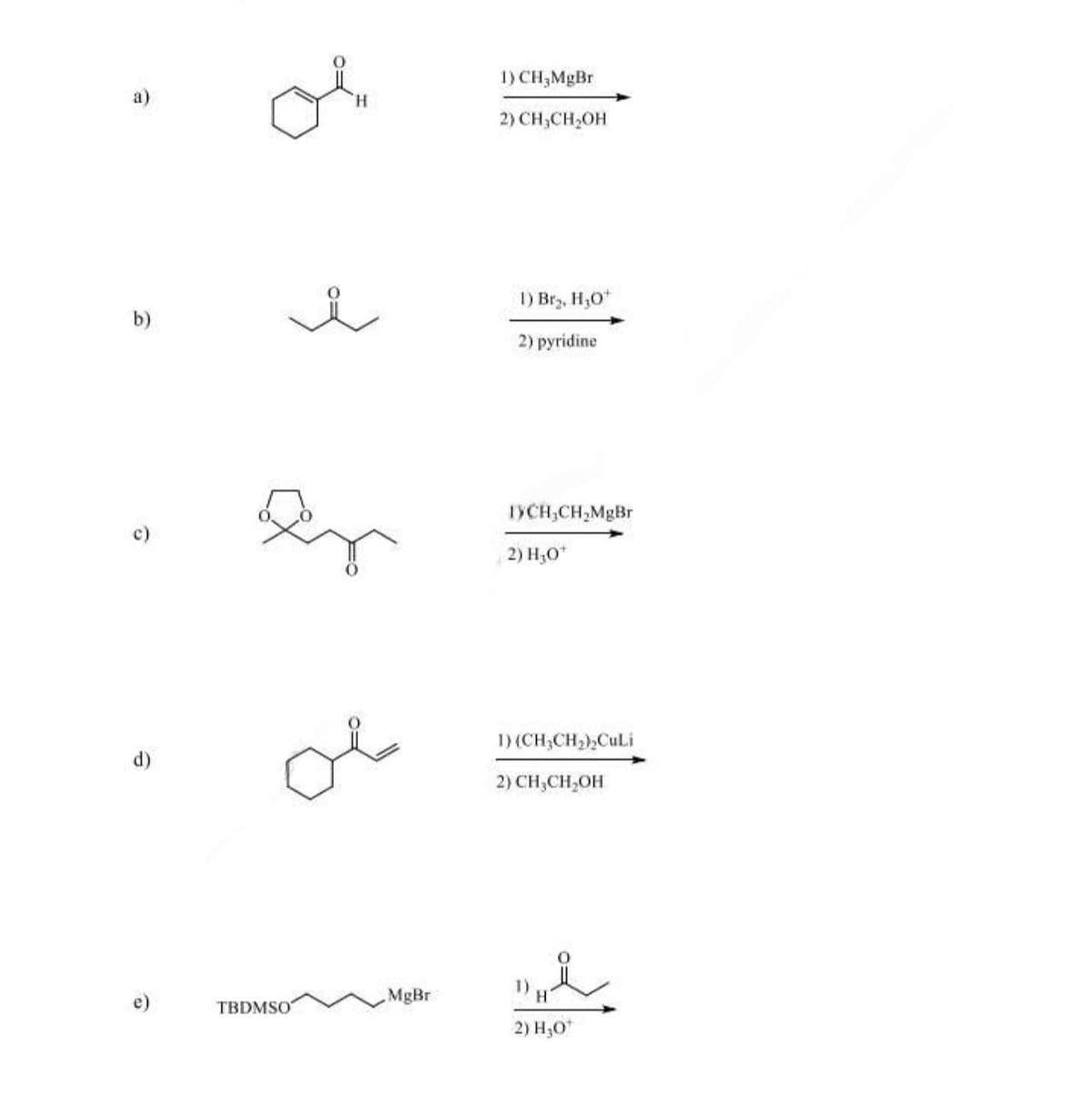
Step by step
Solved in 1 steps with 1 images

- Draw an expanded structural formula of pent-1-en-3-yne/ CH3-CC-CH=CH2 and then label each carbon. Indicate the longest and shortest C-H bond and predict the C—C single bond that has the highest BDE(bond dissociation energy).Calculate ∆Hrxn for the following reaction:N2H4(l) + CH4O(l) → CH2O(g) + N2(g) + 3H2(g)Use the following reactions and given ∆H values.2 NH3(g) → N2H4(l) + H2(g) ΔH = +22.5 kJ/molN2(g) + 3 H 2(g) → 2 NH3(g) ΔH = +57.5 kJ/molCH2O(g) + H2(g) → CH4O(l) ΔH = +81.2 kJ/molHow would you simplify the following expression?
- If you put methylene blue and disperse red 1 mixture into salad dressing, predict what will you observeidk how to solve part b of this problem.one of the chair structures of cis-1-chloro-3-methylcyclohexane is more stable than the other by 15.5 kj/mol (3.7kcal/mo) which is it ? what is the energy cost of a 1,3-diaxial interaction between a chlorine and a methyl group?
- According to the conventions above, what is the sign ( + or ) of the P.E. change (H) for Rxn 3?Calculate delta H for the reaction : Ch4+ 3Cl2= CHCl3+ 3HCl given: CH4 =-74.85(kJ/mol) HCl= -92.30(kJ/mol) CHCl3=-134.3 (kJ/mol) CHCl3=-103.2 (kJ/mol)Ch4+2O2->CO2 +2H2O Do I understand this correctly ? If I burn 1m3/hr of CH4 with 2m3/hr O2, do I get 1m3/hr of CO2 and 2m3/hr of H20? Refering to the above equation.



