Q: Chromate ions react according to the following equation, where the left side of the reaction is yell...
A: Le Chatelier's Principle : is the principle used to predict the effect of a change in conditions on ...
Q: Draw the electron correlation diagram for O2. a. Identify which combinations of atomic orbitals for...
A: We have to draw the molecular orbital diagram of O2
Q: The least reactive carboxylic acid derivative is carboxylate itself. In biological systems, the carb...
A: The acyl phosphate ester acts as a highly reactive acyl derivative like acyl chloride and its reacti...
Q: Which is the reductant and oxidant in the reaction? 2 KMNO4 + 5 C6H806+ 3 H2SO4 – 2 MnS04 + K2SO4 + ...
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and t...
Q: 2. Predict the major product from the following reaction sequence: (Write out complete equations sho...
A:
Q: Which of the following actions would increase foam fractionator efficiency? Lowering salinity (such ...
A: The correct option is as follows:
Q: 7. A 6.0 grams of NaOH is dissolved in distilled water to make 1.60 - liters of solution. What is th...
A:
Q: The Lewis structure of methyl phosphate is shown. Modify the skeletons to show a minor resonance str...
A:
Q: Calculate the percent composition by mass of each element (Pb, C, O) in lead(II) carbonate.
A: Complete solution is given below in next step
Q: Either the reactant (X) or the major organic product is missing from the equation below. Draw the mi...
A: Pd/C will reduce alkenes to alkanes when hydrogen is also present. It’s important to note that the t...
Q: The oxidation number of the central atom Mn2 (C204)3 Answer:
A: To solve this problem we have to calculate the oxidation state of central metal of the given compoun...
Q: 4. What is the concentration of PM10 in air in ppmv if its concentration is 0.15 mg/m3? Assume atmos...
A: Given the concentration of PM10 (molecular weight = 12 g/mol) in air = 0.15 mg/m3. Atmospheric press...
Q: Select the sign of the work based on the change in volume of the cylinder after the reaction.
A: Thermodynamics is the branch of physical chemistry which deals about different forms of energies ...
Q: Which is the reductant and oxidant in the reaction? 2 KMNO4 + 5 C6H8O6 + 3 H2SO4 – 2 MnS04 + K2SO4 +...
A:
Q: According to Kolrausch's law of independent ionic migration, the molar conductivity of an electrolyt...
A:
Q: o.) H;0* р.) 1. SOCI, OH 2. CH;CH,ONa, CH;CH,OH q.) CH3 MCPBA
A:
Q: You add the weighed KHP to a flask containing a 60mL of water rather than 50 mL of water. Explain. Y...
A: Molarity of NaOH is calculated using standard KHP solution(with known concentration) as follows: M...
Q: Calculate the percent composition by mass of each element (C, H, O, P) in methyl phosphate (CH5O4P
A: Determine mass % of each element in methyl phosphate ?
Q: Compare the following: Acid 1: hypochlorous acid, HCIO Acid 2: hydrogen sulfide ion , HS Acid 3: sul...
A:
Q: Circle the most stable conformer 3) Draw the two chair conformations for the compound shown below Cl...
A:
Q: The rate constant for the first-order decomposition at 45 6.2x 10-4 min-1. what is the rate of the r...
A: • The given first order reaction is, 2N2O5 → 4NO2 + O2 • The values provided in the ques...
Q: 5.55 Ammonia is not the only possible fertilizer. Others include urea, which can be produced by the ...
A: The given balanced equation is: CO2(g) + 2NH3(g) -----> CO(NH2)2(s) + H2O(g) In order to find th...
Q: 2. The concentration of of ozone (O3) in Beijing on a summer day (P = 101.325 kPa ; T = 30°C) is 125...
A:
Q: 19. Instructions: MAKE SURE TO CHECK ) 0.4 R REMAINING TIME. On a separate paper form the giver comp...
A: An ionic compound is formed between a metal and nonmetal by the complete transfer of electron from t...
Q: Draw a structural formula for the intermediate in the following reaction: CH3 CHCH2CH3 CH2Cl2 Br2 CH...
A:
Q: 6.8 What mass of butane in grams is necessary to produce 1.5 X 10' kJ of heat? What mass of CO2 is p...
A: The balanced chemical equation for the combustion of methane is, C4H10+132O2→4CO2 + 5H2O 58 g of but...
Q: the decomposition of ammonia to nitrogen and hydrogen gas. Substance S° (J/mol · K) H2(g) 130.7 N2(g...
A: Entropy measures the randomness of the molecule. Gaseous molecule have higher entropy than liquid su...
Q: IM LLI Calculate the standard enthalpy of reaction, AHn, of each of the given acid-base neutralizati...
A:
Q: The pH of a 0.50 M solution of the weak base ethyl amine, C2H5NH2, is 12.20. a. Write the dissociati...
A:
Q: OH
A: 1. Bromination of alkane occurs via radical mechanism. 2. HBr addition reaction in presence of perox...
Q: Draw The molecule morphine with different colours for atom types, proper atomic radius , bind angle ...
A: The structural formula of a molecule furnishes information about the way of bonding between the atom...
Q: For the following reaction, 0.128 moles of potassium hydroxide are mixed with 0.158 moles of phospho...
A: potassium hydroxide + phosphoric acid> potassium phosphate + water 3 KOH + H3PO4 ———> K3PO4 + ...
Q: (d) On the other hand, in quantum physics/mechanics/chemistry, energy is assumed to be quantized and...
A: The speed of a reaction depends on frequency of collision. The more the collision,faster the reactio...
Q: Enter a net ionic equation for the reaction of dilute sulfuric acid with iron. (Assume that sulfuric...
A: • Aqueous sulfuric acid on reaction with solid iron gives aqueous iron (II) sulfate and hydrogen gas...
Q: What is the pH of an aqueous solution of 2.09x10-2M barium hydroxide? pH %3D
A: [Ba(OH)2] = 2.09 x 10-2 M
Q: Liquid octane (CH;(CH,) CH;) reacts with gaseous oxygen gas (02) to produce gaseous carbon dioxide (...
A:
Q: What is the pH of an aqueous solution of 2.09x10-2M barium hydroxide? pH %D
A: Given that, 2.09×10-2 M aqueous solution of barium hydroxide. We have to calculate the pH of the sol...
Q: When 2.00 moles of nitrogen gas and 3.00 moles of oxygen gas were combined in a 4.0 L cylinder (at 3...
A: Number of moles of nitrogen gas = 2 moles Number of moles of oxygen gas = 3 moles Volume of cylinder...
Q: for temperatures in the range 300 erate pressures can be approxima a = 6.15 cal mol- K- and E (a) Ca...
A: Number of moles = 2 moles
Q: A student pipettes 5.00 mL of vinegar into an Erlenmeyer flask, adds indicator and 25 mL of distille...
A: Given, molarity of NaOH = 0.1098 M initial burette reading = 1.35 mL final burette reading = 37.83 m...
Q: Select all of the following elements that are categorized as non-metals. OA. Ag O B. K O C.B O D.S O...
A: Non metals : These are the elements which are placed to right corner of the periodic table and have ...
Q: 9. When heated in solution, 2-pyridinecarboxylic acid(1) loses CO, and forms pyridine. When decarbox...
A:
Q: Define 1. Stoichiometry 2 Formula mass
A: Define 1. Stoichiometry 2 Formula mass
Q: Question 4 What is the cell potential for the following reaction at 298.15 K? Y(s) + z2*(0.010 M) –-...
A:
Q: A balloon filled with helium has a volume of 1.28 × 10³ L at sea level where the pressure is 0.998 a...
A:
Q: Consider the following reaction at equilibrium: CO(g) + H₂O(g) ⇌ CO₂(g) + H₂(g) If CO(g) is removed,...
A:
Q: A buffer solution contains 0.385 M ammonium chloride and 0.444 M ammonia. If 0.0433 moles of hydrobr...
A: Kb for NH3 = 1.8×10-5
Q: 1. Using the Williamson synthesis show how you would prepare 1-ethoxycyclohexane from 1-cyclohexanol...
A: Given, Using the Williamson synthesis show how you would prepare 1-ethoxycyclohexane from 1-cyclohe...
Q: At 3000 K, the partial pressures of A, B and C are 0.6, 0.4 and 0.2 atm, respectively. Calculate A G...
A:
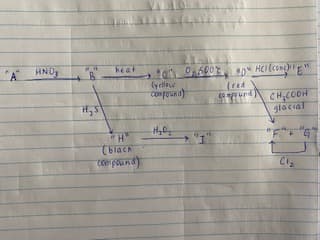
Q: Give structural formulas for A-D in the following synthetic sequence:
A: Ethyl-acetoacetate (EAA) to dicarboxylic acid synthesis : In this conversion first EAA is treate...
Please answer ASAP
Step by step
Solved in 4 steps


