A large jug containing 1.5 kg of water is placed in the freezer. (i) The water cools from 25 °C to 0 °C in a time of 60 minutes. The specific heat capacity of water is 4.2 J / (g °C). Calculate the thermal energy (heat) removed from the water as it cools from 25 °C to 0 °C.
A large jug containing 1.5 kg of water is placed in the freezer. (i) The water cools from 25 °C to 0 °C in a time of 60 minutes. The specific heat capacity of water is 4.2 J / (g °C). Calculate the thermal energy (heat) removed from the water as it cools from 25 °C to 0 °C.
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter17: Energy In Thermal Processes: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 9OQ
Related questions
Question
A large jug containing 1.5 kg of water is placed in the freezer.
(i) The water cools from 25 °C to 0 °C in a time of 60 minutes. The specific heat
capacity of water is 4.2 J / (g °C).
Calculate the thermal energy (heat) removed from the water as it cools from 25 °C
to 0 °C.
Expert Solution
Step 1
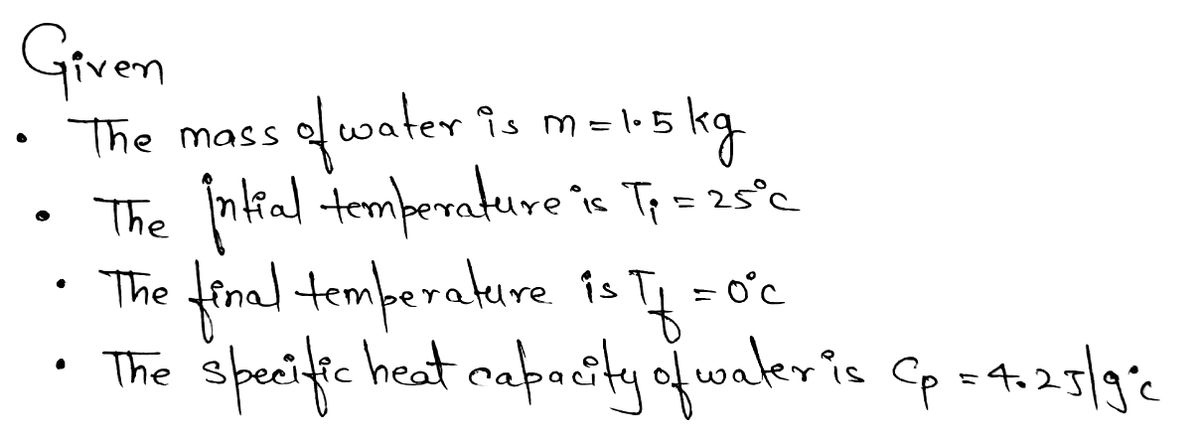
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning