Q: A chemist prepares a solution of vanadium(III) bromide (VBr) by measuring out 0.10 g of VBr3 into a…
A: We have been given mass of solute and volume of solution.We have been asked to to calculate molarity…
Q: Convert 36 kcal to J
A: Given : 36kcal 1 k = 1000 1 cal = 4.184 J Therefore 1kcal = 4184 J
Q: Write the correct empirical formula for hydrogen peroxide, H₂O2
A: Given: molecular formula of hydrogen peroxide is H2O2. As we know, empirical formula of a compound…
Q: 28. A solution is obtained by mixing 10 ml of 0.2 N KOH solution with 30 ml of 0.1 N CH3COOH. The pH…
A: Number of m.moles of KOH in the mixture = (10 x 0.2)= 2Number of m.moles of CH3-COOH in the mixture=…
Q: The conversion of methyl isonitrile into acetonitrile follows first-order kinetics with a half-life…
A:
Q: f 20.0 g of CaCO3 is placed in a 10.0 L reaction vessel at 950oC, what percent of CaCO3 remains in…
A: The reaction for the decomposition of calcium carbonate to form calcium oxide and carbon dioxide…
Q: Write balanced equations for the formation of the following compounds from their elements:…
A: Irion(III) oxide can be formed by the iron and oxygen elements.
Q: The density of titanium is 4.54 g/mL. What is the volume, in milliliters (mL), of 163 g of titanium?…
A: Please find your solution below : Density of a substance is defined as its mass per unit volume. It…
Q: A student dissolves 11. g of glucose (C6H₁2O6) in 500. mL of a solvent with a density of 0.80 g/mL.…
A:
Q: What are the elements that can only hydrogen bond? A. He b. F c. Cl d. O e. C f. N g.p h.…
A: Hydrogen bond : is that bond which is formed between a hydrogen atom and a strong electronegative…
Q: For a given nuclear fission reaction of 235U 235 U+n- →→ ¹4² Ba +⁹Kr +3″ n 92 56 the amount of…
A: Here we have given a nuclear fission reaction of U235 and we are asked to calculate the amount of…
Q: From the data collected for reaction Aproducts determine: a. Order with respect to A b. Rate…
A: We will determine order of reaction first, then using it find rate constant.
Q: A chemist prepares a solution of calcium bromide (CaBr₂) by measuring out 470. g of calcium bromide…
A:
Q: Determine if the organic substrate is oxidized or reduced in the following reaction. Select the…
A: Answer: During the chemical reaction when oxidation number increases then we say that substance has…
Q: Nitric acid readily dissolves UO₂. Why doesn't hydrochloric acid dissolve UO2? Please provide…
A: Nitric acid - HNO3 Uranium dioxide - UO2
Q: Determine the total pressure of a gas mixture that contains gas one at 1.3atm, gas two at 15psi, and…
A: Given --> Gas1 = 1.3 atm Gas2 = 15 psi = 1.021 atm Gas3 = 1480 torr = 1.947 atm
Q: For the following reaction, 17.7 grams of iron are allowed to react with 26.3 grams of hydrochloric…
A: Given data, Mass of iron = 17.7 g Mass of HCl = 26.3 g Data taken from google: Molar mass of Fe =…
Q: You measure 7.47.4 mL of a 50%50% NaOHsolution by weight (its density is 1.53 g mL−1)and dilute it…
A:
Q: 2) Which of the following is a secondary amine? CH₂ NH₂ a) 3) What is the major organic product…
A: The classification of amine is done on the basis of the number of alkyl groups attached to the…
Q: 1 of 15 > The vapor pressure, P, of a certain liquid was measured at two temperatures, T. The data…
A:
Q: 7. Provide reagents for the following transformations LOH гроб pois /^Br B D E Br CI OH Br
A: This problem is based on general organic chemistry reaction mechanism. The reagents are given below.…
Q: Chem 105 Online Density of Metal (Steel) Shot By Displacement 1. Using a weigh boat, add half the…
A: ♦ Density is defined as the mass of a substance per unit volume .∴ The formula of density is -…
Q: 26. Flocculation value of K₂SO4 is much less than that of KBr for Sol A. Floccultion value of CaCl₂…
A: If charge on cation is high then its flocculation value will be low for an anionic sol and if charge…
Q: 7. the normal analytical concentration of K3 Fe(CN)6 8. pK+ for the solution 9. pFe(CN)6 for the…
A: The answer for this problem is provided below in great details within the attached figures.
Q: For each set of isomers, draw the line angle formulas and write the IUPAC name. Draw and…
A:
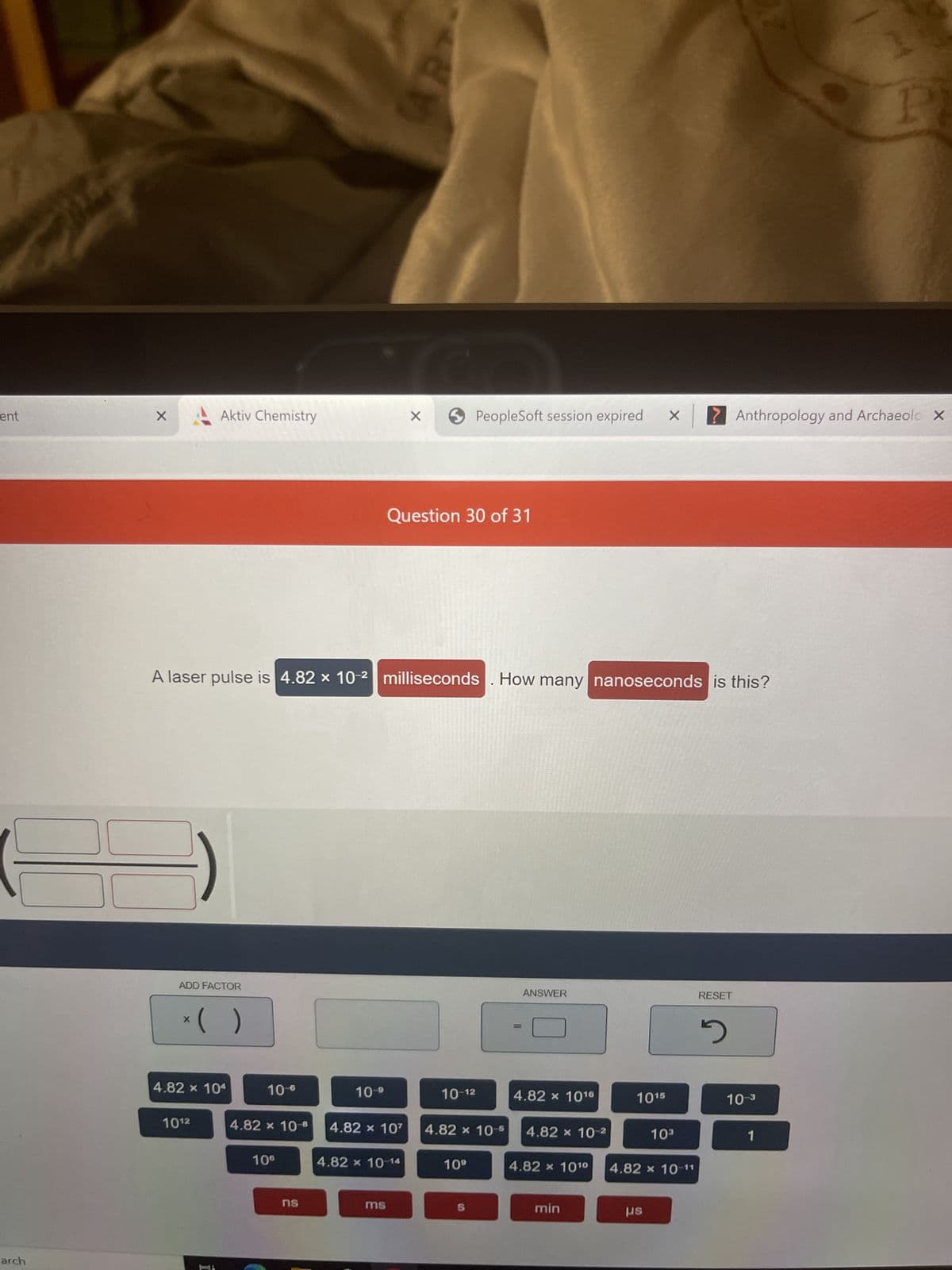
Q: nm HIT ADD FACTOR X 10-6 = 3.50 x 10³ 3.50 x 10² 3.50 spider's silk In 0.001 3.50 x 10-9 nm DELETE…
A: 68. Given,The diameter of a strand of spider's silk is 3.50 micrometers .Required,…
Q: 5. Thermodynamics Calculations. a. When ATP = 3.0 mM, ADP = 2.2 mM and P₁ = 12.1 mM, calculate AG at…
A: The answer for this problem is provided below in great details within the attached figures.
Q: 2. Plot 1/λ (y-axis) vs 1/n2 (x-axis) for hydrogen and attach your graph to the back of this…
A: Solutions- We have to plot the graph of 1/λ (m-1) on Y-axis and 1/n2 on X-axis.
Q: whats the major and minor product $5 ⒸHg (0Ac ) ₂, H₂0 @ NaBHY ●
A:
Q: -23 An oxygen atom has a mass of 2.66 × 10 g and a glass of water has a mass of 0.050 kg. Use this…
A: Given mass of an oxygen atom = 2.66 × 10–23 g And, mas of the glass water = 0.050 kg
Q: 26. Which one of the following complexes exhibits optical isomerism? (a) cis-[PtCl2(NH3)2] (b)…
A: Optical isomers are stereoisomers that are related via non-superimposable mirror images of each…
Q: 37) The most reactive towards nucleophilic acyl substitution is:
A: The reactivity of the acyl compound depends on the partial positive charge present on the carbonyl…
Q: Use the References to access important values if needed for this question. A soda bottle is found to…
A: Consider the given information as follows; The volume of the soda bottle = 772 mL The volume of…
Q: A 23.34 gram sample of chromium is heated in the presence of excess fluorine. A metal fluoride is…
A: we have to calculate the empirical formula of the compound
Q: 6.- How many pi electrons there are in the molecule below?
A: 6) we have to calculate the number of pi electrons in the given 2 structures
Q: Rank the values according to magnitude. 5.39 x 10-5 8.9 × 10² 2.182 x 106 Greatest Least Answer Bank…
A: To rank the values according to the magnitude
Q: Bromination of 2-methylbutane yields predominantly one product with the formula C5H11Br. What is…
A: When 2-methylbutane treated with bromine in presence of sunlight then 2-bromo2-methylbutane is…
Q: -1 Compound X has a molar mass of 579.36 g-mol and the following composition: element phosphorus…
A: Answer: Molecular formula is the type of formula of in which number moles of all the atoms of the…
Q: 1. Determine the number of significant digits in each of the following numbers. a. 6.521 grams d.…
A: Significant figures are the digits of a number that are meaningful in terms of accuracy or…
Q: A chemist prepares a solution of iron(II) bromide (FeBr₂) by measuring out 21. g of iron(II) bromide…
A:
Q: Y Part A What is AS in J/mol-K for this reaction? Express your answer in joules per mole times…
A:
Q: The Lyman series of the hydrogen atom represents transitions from the electronic energy level n to…
A: This question is based on hydrogen spectrum and Bohr's model.
Q: 95. Rank the following alkyl bromide from most reactive to least reactive in a solvolysis reaction…
A: : Solvolysis ∝ stability of carbocation
Q: 8) What is the correct name of the molecule HO a) 5-hydroxy pentanal b) 4-carbonyl-1-butanal c)…
A: given compound
Q: 1. Mass of Metal Shot 2. Initial water volume 3. Final water volume 4. Volume of metal 5. Calculated…
A: Consider the given information as follows;…
Q: A full truckload of garbage has a mass of 26370 pounds. What is this mass in kilograms?
A: we have to covert the value of mass from pounds to kilograms
Q: Each of the following compounds , A-D is equally soluble in the three solvents listed. In each case,…
A: Certain conditions must be met before selecting solvents for recrystallisation. As an example, 'Like…
Q: Which technique would likely be the most useful for distinguishing between the two compounds shown…
A: The NMR spectroscopy is an important tool for the determination of the structure of an organic…
Q: A smart chemical engineering student spilled some peach juice on his expensive T-shirt. He knew that…
A: the two methods are given below
Q: A student runs two experiments with a constant-volume "bomb" calorimeter containing 1300. g of water…
A: We have find out the answer.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- 5.4.5 Chemical microanalysis5.4.5.1 Exploitation of characteristic X-raysElectron probe microanalysis (EPMA) of bulk samples is now a routine technique for obtaining rapid, accurate analysis of alloys. A small electron probe (100 nm diameter) is used to generate X-rays from a defined area of a polished specimen and the intensity of the various characteristic X-rays measured usingeither wavelength-dispersive spectrometers (WDS) or energy-dispersive spectrometers (EDS). Typically the accuracy of the analysis is š0.1%. One of the limitations of EPMA of bulk samples is that the volume of the sample which contributes to the X-ray signal is relatively independent of the size of theelectron probe, because high-angle elastic scattering of electrons within the sample generates X-rays. The consequence of this is that the spatial resolution of EPMA is no better than ¾2 µm. In the last few years EDX detectors have been interfaced totransmission electron microscopes which are capable of operating…Describe the origin of shot noise in a spectrophotometer. How does the relativeuncertainty vary with concentration if shot noise is the major noise source?Calculate the Ksp for Ni(OH)2 Ni(OH)2(s) + 2 e- ⟶ Ni(s) + 2OH-(aq); Eo = -0.720 VNi2+(aq) + 2 e- ⟶ Ni(s); Eo = -0.250 V 7.56x1015 1.15x10-8 1.70x10-33 1.32x10-16
- using the following information to construct a valid hypothesis addressing the effect of pH and temperature on the hydrolysis of starch. Per bench, prepare the stock amylase enzyme solution by adding distilled water to the powdered enzyme in the flask , each bench will share one flaks of enzyme solution. per groups, turn on the spectrometer and set the wavelength to 560nm.allow the instruments to warm up for at least 10 minutes. per group, set up 17 cuvettes in a Styrofoam rack. Add 3 drops of iodine (IKI) to each coveter. per group, prepare the “black” cuvette by rapidly adding 3 mL of maltose standard solution to one of The prepared cuvettes containing IKI. Rapid addition of the maltose will mix the maltose and IKIA 1.2cm diameter glass cuvette is filled with a Cu(NH3)42+ solution that is deep blue. The cuvette is placed into a pre-blanked spectrophotometer. At 620nm, the transmittance is 23%. If the solution has molarity = 0.0133 M and path length of light through the solution = 1.2 cm, what is the value of molar absorptivity at 620 nm?1.How many milliliters of 100 % (w/v) ethanol are equal to 0.57 millimoles (mmole)? (Hint: It is not 57 ml.) 2. What is the molar concentration, millimolar (mM) concentration, and micromolar (µM) concentration when 3 micromoles (µmoles) of glycine are diluted to a final volume of 6 L? 3. If the percent transmittance of a colored solution is found to be 35%, what amount of distilled water must be added to 100 ml of this solution in order to have a solution with a percent transmittance of 75%.
- Answer Q45 & 46 show-all-working-explaining-detailly-each-step Answer should be typewritten using a computer keyboard.The resolution product of Ag2SO3 is 1.5 x 10-14. Calculate E0 for Ag2SO3 (k) + 2e- ↔ 2 Ag (k) + SO32-. Ag + + e- ↔ Ag E0 = + 0.799 V A. + 0.799 VB. + 0.803 VC. + 0.020 VD. + 0.390 Vplease quickly ,within 1hour thanks ! If it takes longer than an hour, i don't need the answer (1) List the detectors ofHPLC. Describe the advantage and disadvantage for two wideused detectors
- What is the frequency of the xray with a wavelength of 1.0 x 10 to the negative 10th poerThe Ksp of Zn(OH)2 is 3.00 x 10^-17Why is the blank solution needed in spectrophotometry? A. A blank solution is used to clean the spectrophotometer b. A blank solution is used to calibrate the spectrophotometer readings: they document the baseline response of the environment- instrument- sample system



