A metal alloy has a grain diameter of 1.7x102 mm. After a heat treatment at 450°C for 250 minutes, the grain diameter has increased to 4.5x102 mm. Compute the time required (in minutes) for a specimen of this same material (with do = 1.7x102 mm) to achieve a grain diameter of 8.7x102 mm while being heated at 450°C. Assume that n = 2. Your final answer should be rounded to the nearest ones place (e.g. no decimals). Answer:
A metal alloy has a grain diameter of 1.7x102 mm. After a heat treatment at 450°C for 250 minutes, the grain diameter has increased to 4.5x102 mm. Compute the time required (in minutes) for a specimen of this same material (with do = 1.7x102 mm) to achieve a grain diameter of 8.7x102 mm while being heated at 450°C. Assume that n = 2. Your final answer should be rounded to the nearest ones place (e.g. no decimals). Answer:
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter3: Molecules, Moles, And Chemical Equations
Section: Chapter Questions
Problem 3.108PAE: 3.108 As chip speeds increase, the width of the interconnects described in Problem 3.107 must be...
Related questions
Question
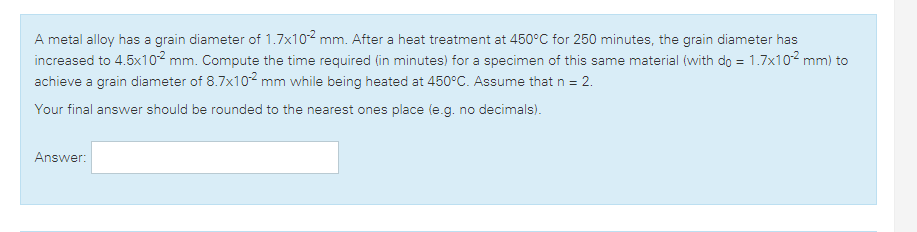
Transcribed Image Text:A metal alloy has a grain diameter of 1.7x102 mm. After a heat treatment at 450°C for 250 minutes, the grain diameter has
increased to 4.5x102 mm. Compute the time required (in minutes) for a specimen of this same material (with do = 1.7x102 mm) to
achieve a grain diameter of 8.7x102 mm while being heated at 450°C. Assume that n = 2.
Your final answer should be rounded to the nearest ones place (e.g. no decimals).
Answer:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole