A normal person contains about 1.4 gallons of blood and has a sodium blood level (or sodium concentration) of about 140 milliequivalents per liter. Calculate the total grams of sodium that are dissolved in a normal persons blood.
A normal person contains about 1.4 gallons of blood and has a sodium blood level (or sodium concentration) of about 140 milliequivalents per liter. Calculate the total grams of sodium that are dissolved in a normal persons blood.
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter8: Solutions
Section: Chapter Questions
Problem 8.41EP
Related questions
Question

Transcribed Image Text:A normal person contains about 1.4 gallons of blood and has a sodium blood
level (or sodium concentration) of about 140 milliequivalents per liter.
Calculate the total grams of sodium that are dissolved in a normal persons
blood.
Expert Solution
Step 1
The numerical conversion factor may be explained as the mathematical factor that is used to transfer to other units and also establishes the relationship linking the two units.
Step 2
The expression to calculate the grams of the sodium that are dissolved in blood is shown below:
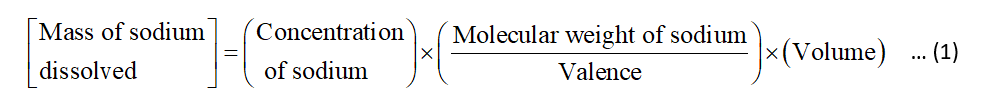
The value of the concentration of sodium is 140 meq/L.
The value of the valence is 1.
The value of the volume is 1.4 gallons.
The value of the molecular weight is 23 g/mol.
Substitute the value in equation (1)
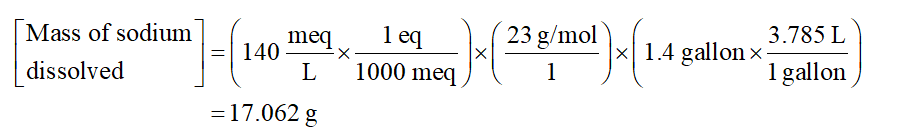
Therefore, the value of the grams of the sodium that are dissolved in blood is 17.062 g.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co