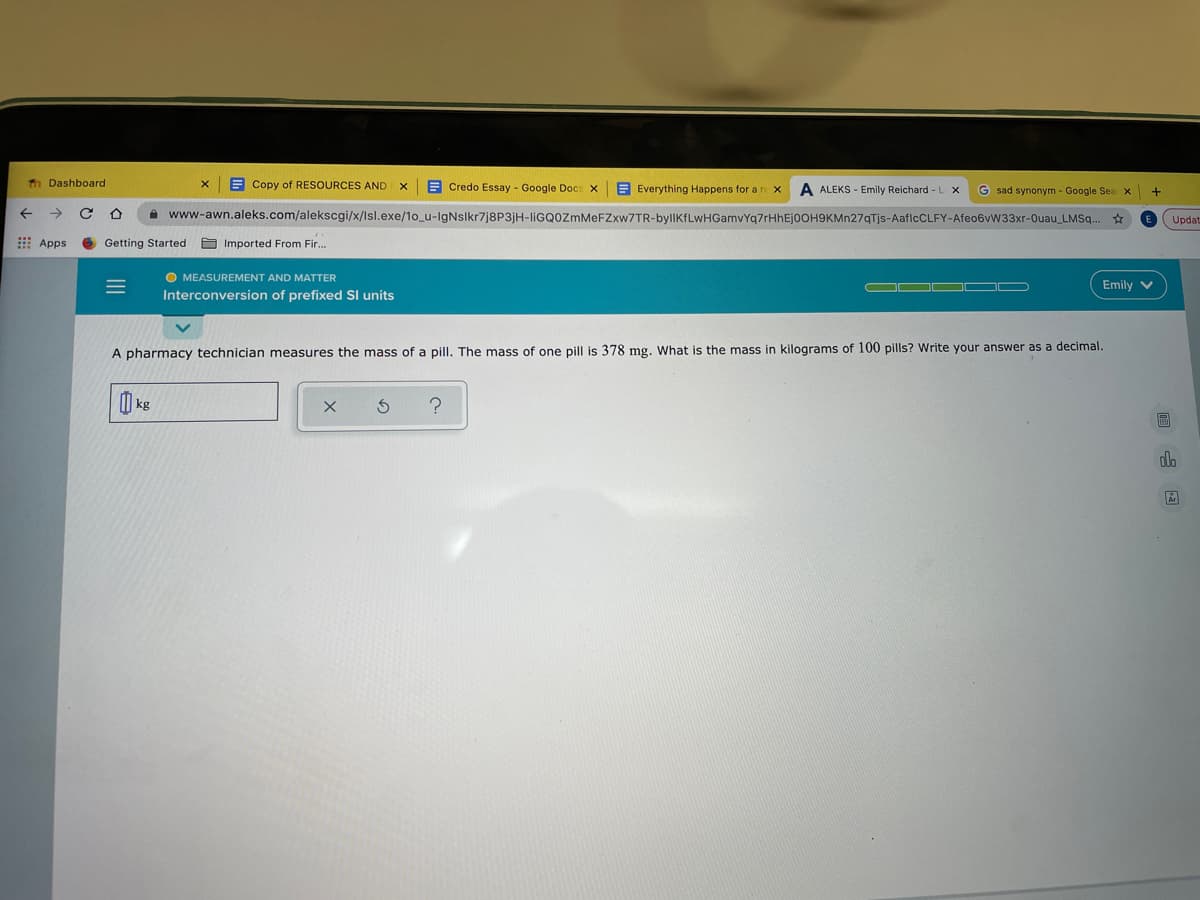
A pharmacy technician measures the mass of a pill. The mass of one pill is 378 mg. What is the mass in kilograms of 100 pills? Write your answer as a decimal. | kg
A pharmacy technician measures the mass of a pill. The mass of one pill is 378 mg. What is the mass in kilograms of 100 pills? Write your answer as a decimal. | kg
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter34: Particle Size Determination
Section: Chapter Questions
Problem 34.12QAP
Related questions
Question
Transcribed Image Text:n Dashboard
E Copy of RESOURCES AND
A Credo Essay - Google Docs X
E Everything Happens for a re x
A ALEKS - Emily Reichard - L X
G sad synonym - Google Sea x
->
A www-awn.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IIGQ0ZMMEFZXW7TR-byllKfLwHGamvYg7rHhEj0OH9KMn27qTjs-AaflcCLFY-Afeo6vW33xr-Ouau LMSq.
(Updat
E Apps
Getting Started
O Imported From Fir..
O MEASUREMENT AND MATTER
Interconversion of prefixed Sl units
Emily v
A pharmacy technician measures the mass of a pill. The mass of one pill is 378 mg. What is the mass in kilograms of 100 pills? Write your answer as a decimal.
W kg
do
II
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
