Q: Why is it necessary to specify the temperaturewhen making a table listing ∆G values?
A: ∆G is defined the change of Gibbs (free) energy for a system; ∆G° is the Gibbs energy change for a…
Q: 1. SEP Compare Data Of the metals shown in the figure, which has the highest melting point? Which…
A: Of the metals given in the figure, molybdenum (Mo) has the highest melting temperature i.e. 2617°C
Q: What are some U.S. units that are used to measure each of the following? A. length B. volume C.…
A: In the United States, the imperial system of measurement is followed. These units of measurements…
Q: volumetric flask made of Pyrex glass is calibrated at 20 °C.It is filled to the 100 mL mark with 35…
A: Original volume is 100 mL. Delta T = 20 - 35 = -15°C After cooling to 20 °C, the volume is Vfinal=…
Q: What do you call to the unit amount of a substance used for measuring large quantities of very small…
A: The mole (particularly millimoles and molarity) is a commonly used unit in medicine. For example,…
Q: What is the isoelectric point of AA? [Select]
A: Isoelectric point (pI) of amino acid: Isoelectric point is the pH at which amino acid shows no…
Q: Which pharmaceutical products need specific gravity measurement? Mention three examples
A: Pharmacy is the branch of health science that connects medical science with chemistry. It is…
Q: Now calculate the slope and equation for the best fit line. You may use excel
A:
Q: Convert between mass and moles using the molar mass of a substance.
A: Molarity is defined by a number of moles of a substance in a solution (in litres). Molar mass is…
Q: Which log is used to measure porosity accurately
A: Sonic logging equipment provided the first porosity analyses, which were accessible in the 1950s.…
Q: Determine the following equivalents: (Round to the nearest tenth on weight 6.8 oz = 7.2 tsp = 8. 45…
A: To convert one unit to another, we must find the value of unit in comparison to 1 of the second…
Q: föllowing would dispersion forces be the most important factor in determining physical properties in…
A: The London dispersion forces are defined as the weakest existing intermolecular forces. It is a…
Q: For the table below, specify the dominant intermolecular force involved for each substance in the…
A: Intermolecular forces along with bonds determine the boiling points of a compound as the more the…
Q: Calculate the surface area to volume ratio of a cube 5mm and a cube 9mm.
A: Biological processes are processes that are crucial for all living organism to live and are composed…
Q: if you have Quinoa Serving size -170 grams Carbohydrates – 109 grams calculate the DV % for these…
A: DV% is how much of a nutrient is present in a single serving of a packaged food or a dietary…
Q: If 65 percent of your body mass is water, determine how many kilograms of water you contain.
A: Water is the main component of the human body and it forms nearly 60% of body mass. Different organs…
Q: Compare/Contrast elemental and ionic lead in chemical & physical properties and their effect on…
A: Lead(Pb): It is a chemical element with the symbol Pb. It is amphoteric which is why it shows weak…
Q: Note: answer only five questions On what basis are the materials properties categorized? Give…
A: Materials - Material is defined as a substance. It can be made up from substance or from group of…
Q: If a 0.2g of oil consumed 1ml of sodium thiosulphate, calculate its Iodine value and classify the…
A: Lipids are classified into fats, oil, and wax. Fats are also called triglycerides, where three fatty…
Q: For a powder binding process that mixes a 1:2 volume ratio mixture of semi-crystalline polyethylene…
A: Given: Density of semi-crystalline substance = 0.900 g/cm3 Density of crystalline substance = 0.998…
Q: determine the density of a metal that has a mass of 2.17g and a volume of 0.35 cm^3
A: The unit of density is gm/cm3 .
Q: 4. A bilayer polymer slab (1.2m long, 1.4m wide, 5 cm thick) is placed on a hot plate, which keeps…
A: This answer would have solved so nicely by handwritten note but you aksed for digitally so here you…
Q: Calculate the solubility of Ag,CO3 (in mol/L) at 25 °C in a 0.02 M Na,CO3 solution Hint: Ag,CO3(s) →…
A: Solubility equilibrium is a type of dynamic equilibrium that exists when a chemical compound in the…
Q: Name and strength for three pharmaceutical products that its specific gravity should be measured ?
A: Pharmaceutical product or drugs are the compounds that are used to prevent, treat, cure, or diagnose…
Q: the vapor phase concentration in ppm(v)?
A: Vapour phase concentration is the amount of water vapor present in a unit volume of air, usually…
Q: Suppose a certain liquid in contact with a surface has cohesive and adhesive forces such that its…
A: Cohesion and Adhesion are macroscopic properties of a liquid. These forces act on the surface when…
Q: Explain briefly effects and importance of following phenomena on bioreactor design. oxygen demand…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: If 5.0 g of calcium hydroxide are needed, how many moles of magnesium hydroxide would be produced?…
A: 1.27 Moles of Mg(OH)2 are produced from 1 mole of Ca(OH)2.
Q: BSA (mg/ml) Absorbancy 595nm 0.225 1 0.310 2 0.420 3 0.510 4 0.610 0.720 6 0.810 7 0.915 8 0.950…
A: The Bradford Assay is is a sensitive assay to detect and quantify protein in solution.
Q: Calculate the molarity (M) of a 65% (w/v) copper sulfate solution (m.w. = 160.0 g/mol).
A: The molarity of a solution is defined as moles of solute in per litre of solution. To calculate…
Q: 05:Calculate the grams of each substance requied to prepare the following solutions:(a)250 ml of 0.1…
A: Multiple questions asked. 1st question will be answered, as permissible by guidelines. Asked :…
Q: 33. Which graph best represents the following data? A, B, C or D Height (cm) Weight (kg) TT 150 45…
A: INTRODUCTION There is an increase in weight according to the increase in height.
Q: mass Density volume Mass density x volume %3D 3. Solve the following word problem using the formula…
A: Density is defined as mass per unit volume. The average density of a substance is calculated by…
Q: A slice of pizza has 500 kcal. If we could burn the pizza and useall the heat to warm a 50-L…
A: Specific heat is a physical property of substance. It is the amount of heat needed per unit mass to…
Q: Define the mole, and calculate the molar mass of a compound from the molecular formula.
A: The mass of substance present in a sample of a given compound is divided upon the amount of…
Q: Explain the principles behind the melting curve and how this helps determine G+C content. Include an…
A: Melting curve inspection is an evaluation of the dissociation features of double stranded DNA, in…
Q: A solution of calcium phosphate in water contain 235 gms of CP per L at 30 deg c. The density of the…
A: Percentage composition is calculated as the percentage by mass of individual elements contributed to…
Q: Compute BMI using metric measurements Height: 162.5 cm (5'4) Weight: 47 kg
A: Body mass index depends on the height and body weight of an individual. It is linked to various…
Q: 77 10 Comparison c
A: The synthesis of protein is through a process called translation of mRNA, which has been transferred…
Q: Using the figure, answer the following questions.a. What is the molar composition of the vapor in…
A: The horizontal lines denote constant temperature. The curve at point x denotes liquid having mole…
Q: Calculate the plane density of (110) plane at BCC sbructure Crystal : 2
A: For this (110) plane there is one atom at each of the four cube corners through which it passes,…
Q: (b) Define the melting temperature (Tm) of a polymer and describe a method that could be used to…
A: The melting point is determined as the temperature at which a polymer transits from the crystalline…
Q: Check this 1 1 1 1 1 deciliter centiLiter milliLiter microLiter nanoliter (dL) (CL) (mL) (mL) (NL)…
A: Introduction A unit is a numerical way of expressing physical quantity such as weight, length, and…
Q: ounce 8 = how many drams ?
A: The different systems of measurement used in medicine include the household system, the avoirdupois…
Q: GC/L what does the E and GC/L stand for in water measurement?
A: EOP - electro-osmotic-pulse GCL - geosynthetic liners
Q: Give some characteristics of ground substance.
A: The tissue that connects and provides support to all remaining body tissues is called connective…
Q: If a liter of a cough syrup weighs 1285g, what is the specific gravity?
A: Specific gravity can be defined as the ratio of the substance's weight to the weight of the equal…
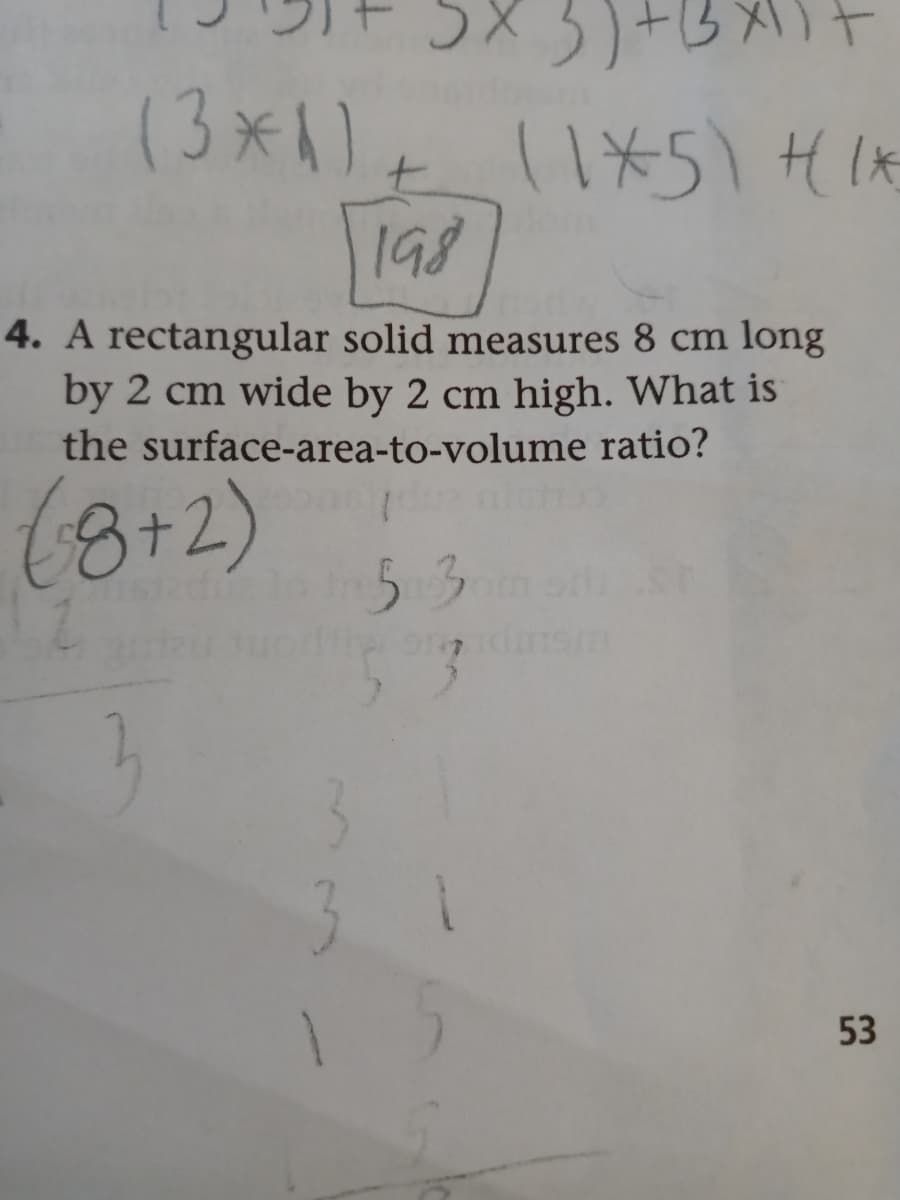
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What is the distance between G&R, G&L, R&L8.5gm/250ml = (x)gm/630mlQ.1. A solution of calcium phosphate in water contain 235 gms of CP per L at 30 deg c. The density of the substance at this temperature is 1.143 gm/cc. Calculate , composition in wt%, mole%,atomic % and volume%of water. Density of water 0.999 gm/cc.



