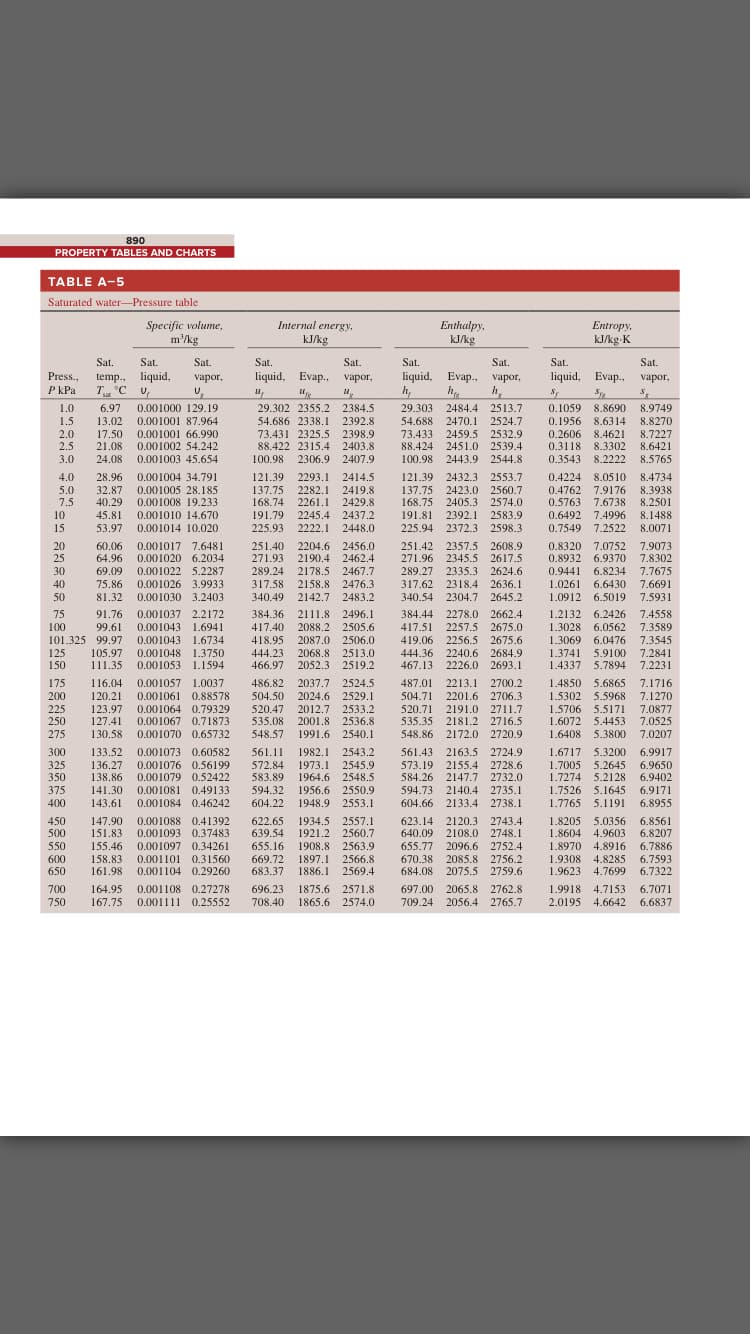
A rigid 14-L vessel initially contains a mixture of liquid water and vapor at 100°C with 12.3 percent quality. The mixture is then heated until its temperature is 180°C. The final state is superheated water and the internal energy at this state should be obtained by interpolation. Calculate the heat transfer required for this process. Use data from the steam tables. Water 100°C x = 0.123 The heat transfer required for this process is kJ.
A rigid 14-L vessel initially contains a mixture of liquid water and vapor at 100°C with 12.3 percent quality. The mixture is then heated until its temperature is 180°C. The final state is superheated water and the internal energy at this state should be obtained by interpolation. Calculate the heat transfer required for this process. Use data from the steam tables. Water 100°C x = 0.123 The heat transfer required for this process is kJ.
Elements Of Electromagnetics
7th Edition
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Sadiku, Matthew N. O.
ChapterMA: Math Assessment
Section: Chapter Questions
Problem 1.1MA
Related questions
Question
Transcribed Image Text:890
PROPERTY TABLES AND CHARTS
TABLE A-5
Saturated water–Pressure table
Specific volume
m'/kg
Internal energy,
Enthalpy,
kJ/kg
Entropy,
kJ/kg-K
kJ/kg
St.
liquid, Evap., vapor,
h,
Sat.
Sat.
Sat.
Sat.
Sat.
Sat.
Sat.
St.
Press.,
liquid, Evap., vapor,
temp., liquid,
T "C v,
vapor,
liquid, Evap.,
vapor,
P kPa
U,
1.0
6.97
13.02
0.001000 129.19
29.302 2355.2 2384.5
29.303 2484.4
2513.7
2524.7
0.1059
8.8690
8.9749
8.8270
8.7227
8.6421
1.5
0.001001 87.964
54.686 2338.1 2392.8
54.688
2470.1
0.1956
8.6314
2.0
2.5
17.50
21.08
0.001001 66.990
0.001002 54.242
73.431 2325.5 2398.9
88.422 2315.4 2403.8
73.433 2459.5 2532.9
88.424
0.2606 8.4621
2451.0 2539.4
0.3118 8.3302
3.0
24.08
0.001003 45.654
100.98
2306.9 2407.9
100.98
2443.9
2544.8
0.3543 8.2222 8.5765
4.0
28.96
0.001004 34.791
0.001005 28.185
0.001008 19.233
121.39 2293.1 2414.5
121.39 2432.3 2553.7
0.4224 8.0510
8.4734
5.0
7.5
32.87
40.29
137.75
168.74
2282.1 2419.8
2261.1 2429.8
137.75
168.75 2405.3
2423.0 2560.7
2574.0
0.4762 7.9176
0.5763 7.6738
8.3938
8.2501
10
45.81
53.97
0.001010 14.670
191.79 2245.4 2437.2
191.81
2392.1
2583.9
0.6492 7.4996
8.1488
15
0.001014 10.020
225.93
2222.1
2448.0
225.94 2372.3 2598.3
0.7549 7.2522
8.0071
20
25
60.06
64.96
0.001017 7.6481
0.001020 6.2034
251.40
271.93
2204.6 2456.0
2190.4 2462.4
251.42
271.96 2345.5
2357.5 2608.9
2617.5
0.8320 7.0752
0.8932 6.9370
7.9073
7.8302
30
69.09
0.001022 5.2287
289.24 2178.5 2467.7
289.27
2335.3 2624.6
0.9441 6.8234
7.7675
40
75.86
0.001026 3.9933
317.58
2158.8 2476.3
317.62 2318.4 2636.1
1.0261 6.6430
7.6691
50
81.32
0.001030 3.2403
340.49
2142.7 2483.2
340.54
2304.7
2645.2
1.0912 6.5019
7.5931
75
91.76
0.001037 2.2172
384.36 2111.8
2496.1
384.44
2278.0 2662.4
1.2132 6.2426
7.4558
100
0.001043 1.6941
99.61
101.325 99.97
105.97
111.35
417.40
2088.2 2505.6
417.51
2257.5 2675.0
1.3028 6.0562
7.3589
0.001043 1.6734
418.95
2087.0 2506.0
419.06 2256.5 2675.6
1.3069 6.0476
7.3545
1.3741
125
150
0.001048 1.3750
0.001053 1.1594
444.23
466.97
2068.8 2513.0
444.36
467.13
2240.6
2684.9
2226.0 2693.1
5.9100
7.2841
7.2231
2052.3 2519.2
1.4337 5.7894
175
116.04
0.001057 1.0037
486.82 2037.7 2524.5
487.01
2213.1
2700.2
1.4850 5.6865 7.1716
200
120.21
0.001061 0.88578
504.50
2024.6 2529.1
504.71
2201.6 2706.3
1.5302 5.5968
7.1270
225
250
123.97
127.41
130.58
0.001064 0.79329
520.47
535.08
548,57
2012.7 2533.2
2001.8 2536.8
520.71
2191.0
2711.7
2181.2 2716.5
1.5706 5.5171
7.0877
0.001067 0.71873
535.35
1.6072 5.4453
7.0525
275
0.001070 0.65732
1991.6 2540.1
548.86 2172.0 2720.9
1.6408 5.3800
7.0207
300
133.52
0.001073 0.60582
561.11
1982.1 2543.2
561.43 2163.5 2724.9
1.6717 5.3200
6.9917
325
350
136.27
138.86
0.001076 0.56199
0.001079 0.52422
572.84
583.89
1973.1 2545.9
1964.6 2548.5
573.19 2155.4
584.26 2147.7
2728.6
2732.0
1.7005 5.2645
1.7274 5.2128
6.9650
6.9402
141.30
1.7526 5.1645
1.7765 5.1191
375
0.001081 0.49133
594.32
604.22
1956,6
2550.9
594.73 2140.4 2735.1
6.9171
400
143.61 0.001084 0.46242
1948.9 2553.1
604.66 2133.4 2738.1
6.8955
450
500
147.90
151.83
0.001088 0.41392
0.001093 0.37483
622.65
639.54
1934.5 2557.1
1921.2 2560.7
623.14 2120.3 2743.4
640.09
655.77
670.38
684.08
1.8205 5.0356
1.8604 4.9603
6.8561
6.8207
2108.0 2748.1
550
155.46
0.001097 0.34261
655.16
1908.8 2563.9
2096.6 2752.4
1.8970 4.8916
6.7886
6.7593
6.7322
0.001101 0.31560
600
650
158.83
161.98 0.001104 0.29260
669.72 1897.1 2566.8
1886.1
2085.8
2075.5
2756.2
2759.6
1.9308 4.8285
1.9623
4.7699
683.37
2569.4
697.00
709.24
700
164.95
0.001108 0.27278
696.23
1875.6 2571.8
2065.8 2762.8
1.9918 4.7153 6.7071
750
167.75
0.001111 0.25552
708.40
1865.6 2574.0
2056.4
2765.7
2.0195 4.6642
6.6837

Transcribed Image Text:A rigid 14-L vessel initially contains a mixture of liquid water and vapor at 100°C with 12.3 percent quality. The mixture is then heated
until its temperature is 180°C. The final state is superheated water and the internal energy at this state should be obtained by
interpolation. Calculate the heat transfer required for this process. Use data from the steam tables.
Water
100°C
x = 0.123
The heat transfer required for this process is
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:
9781118170519
Author:
Norman S. Nise
Publisher:
WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:
9781337093347
Author:
Barry J. Goodno, James M. Gere
Publisher:
Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:
9781118807330
Author:
James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:
WILEY