A sample of iron ore is dissolved in acid, and the iron is converted to Fe2+. The sample is then titrated with 47.20 mL of 0.02240 M MnO4¯ solution. The oxidation-reduction reaction that occurs during titration is as follows: MnO4(aq) + 5FE2*(aq) + 8H*(aq) 4H2O Mn2*(aq) + 5FE3+ (aq) + How many moles of MnO4 were added to the solution? Blank 1 How many moles of Fe 2+ were in the sample? Blank 2 How many grams of iron were in the sample? Blank 3 If the sample had a mass of 0.8890 g, what is the percentage of iron in the sample? Blank 4
A sample of iron ore is dissolved in acid, and the iron is converted to Fe2+. The sample is then titrated with 47.20 mL of 0.02240 M MnO4¯ solution. The oxidation-reduction reaction that occurs during titration is as follows: MnO4(aq) + 5FE2*(aq) + 8H*(aq) 4H2O Mn2*(aq) + 5FE3+ (aq) + How many moles of MnO4 were added to the solution? Blank 1 How many moles of Fe 2+ were in the sample? Blank 2 How many grams of iron were in the sample? Blank 3 If the sample had a mass of 0.8890 g, what is the percentage of iron in the sample? Blank 4
Chapter4: Types Of Chemical Reactions And Solution Stoichiometry
Section: Chapter Questions
Problem 109AE: The blood alcohol (C2H5OH) level can be determined by titrating a sample of blood plasma with an...
Related questions
Question
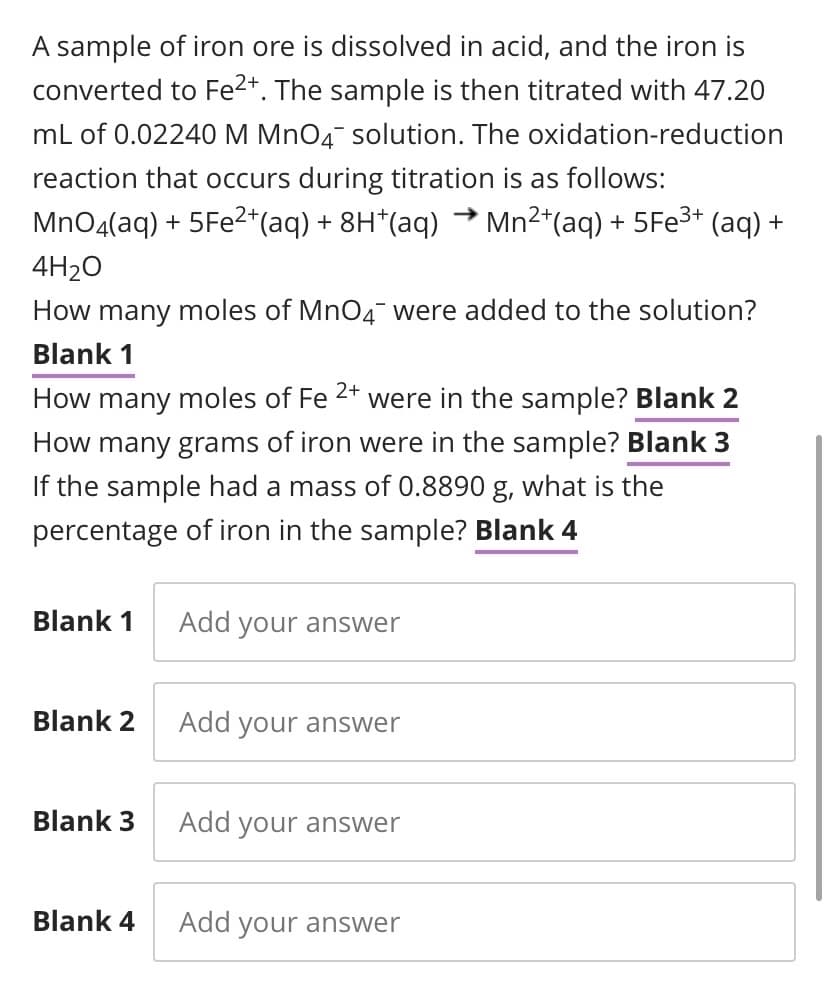
Transcribed Image Text:A sample of iron ore is dissolved in acid, and the iron is
converted to Fe2*. The sample is then titrated with 47.20
mL of 0.02240 M MnO4 solution. The oxidation-reduction
reaction that occurs during titration is as follows:
MnO4(aq) + 5FE2*(aq) + 8H*(aq) → Mn²*(aq) + 5FE3* (aq) +
4H2O
How many moles of MnO4¯ were added to the solution?
Blank 1
2+
How many moles of Fe
were in the sample? Blank 2
How many grams of iron were in the sample? Blank 3
If the sample had a mass of 0.8890 g, what is the
percentage of iron in the sample? Blank 4
Blank 1
Add your answer
Blank 2
Add your answer
Blank 3
Add your answer
Blank 4
Add your answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning