A saturated solution of Ag2CO3 is made: Ag2CO3(s) + 2Ag*(aq) + CO32 (aq) What happens to the equilibrium when some more solid Ag2CO3 is added to the solution? The equilibrium shifts to the right The equilibrium shifts to the left. The equilibrium shifts in the direction of dissolving more solid. O Nothing happens, the solution is already saturated.
A saturated solution of Ag2CO3 is made: Ag2CO3(s) + 2Ag*(aq) + CO32 (aq) What happens to the equilibrium when some more solid Ag2CO3 is added to the solution? The equilibrium shifts to the right The equilibrium shifts to the left. The equilibrium shifts in the direction of dissolving more solid. O Nothing happens, the solution is already saturated.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter15: Complex Ion And Precipitation Equilibria
Section: Chapter Questions
Problem 38QAP: To a beaker with 500 mL of water are added 95 mg of Ba(NO3)2, 95 mg of Ca(NO3)2, and 100.0 mg of...
Related questions
Question
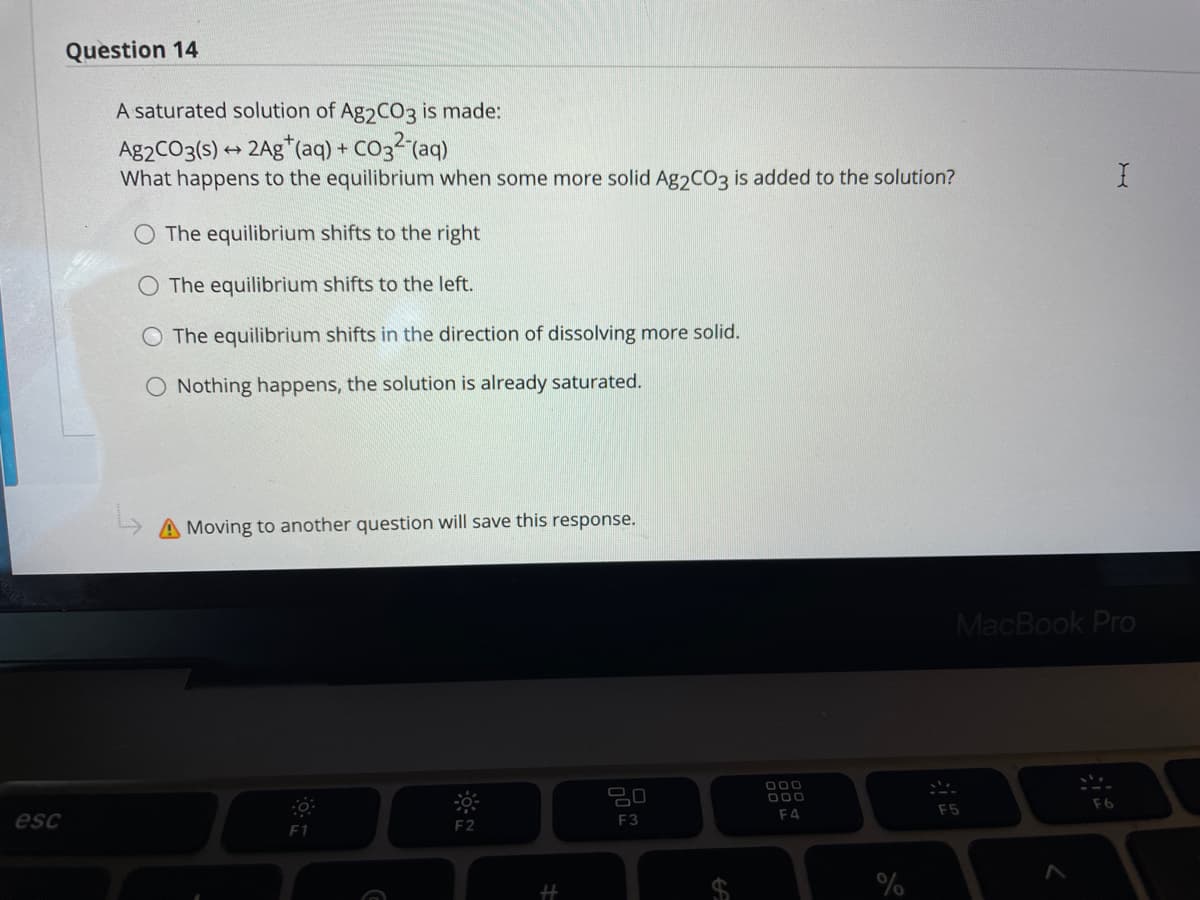
Transcribed Image Text:Question 14
A saturated solution of Ag CO3 is made:
Ag2CO3(s) + 2Ag*(aq) + CO32(aq)
What happens to the equilibrium when some more solid Ag2CO3 is added to the solution?
The equilibrium shifts to the right
The equilibrium shifts to the left.
The equilibrium shifts in the direction of dissolving more solid.
O Nothing happens, the solution is already saturated.
A Moving to another question will save this response.
MacBook Pro
000
D00
F6
F4
F5
esc
F2
F3
F1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning