A solution is prepared by adding 500 mL of 0.3 M NaCIO to 500 mL of 0.4 M HCIO. What is the pH of this solution? Multiple Choice The pH will be equal to the pKa of hypochlorous acid. The pH will be less than the pKą of hypochlorous acid. The pH will be greater than the pka of hypochlorous acid. The pH will equal the pKþ of sodium hypochlorite. None of thece choices are corrert
A solution is prepared by adding 500 mL of 0.3 M NaCIO to 500 mL of 0.4 M HCIO. What is the pH of this solution? Multiple Choice The pH will be equal to the pKa of hypochlorous acid. The pH will be less than the pKą of hypochlorous acid. The pH will be greater than the pka of hypochlorous acid. The pH will equal the pKþ of sodium hypochlorite. None of thece choices are corrert
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter13: Acids And Bases
Section: Chapter Questions
Problem 188IP: Isocyanic acid (HNCO) can be prepared by heating sodium cyanate in the presence of solid oxalic acid...
Related questions
Question
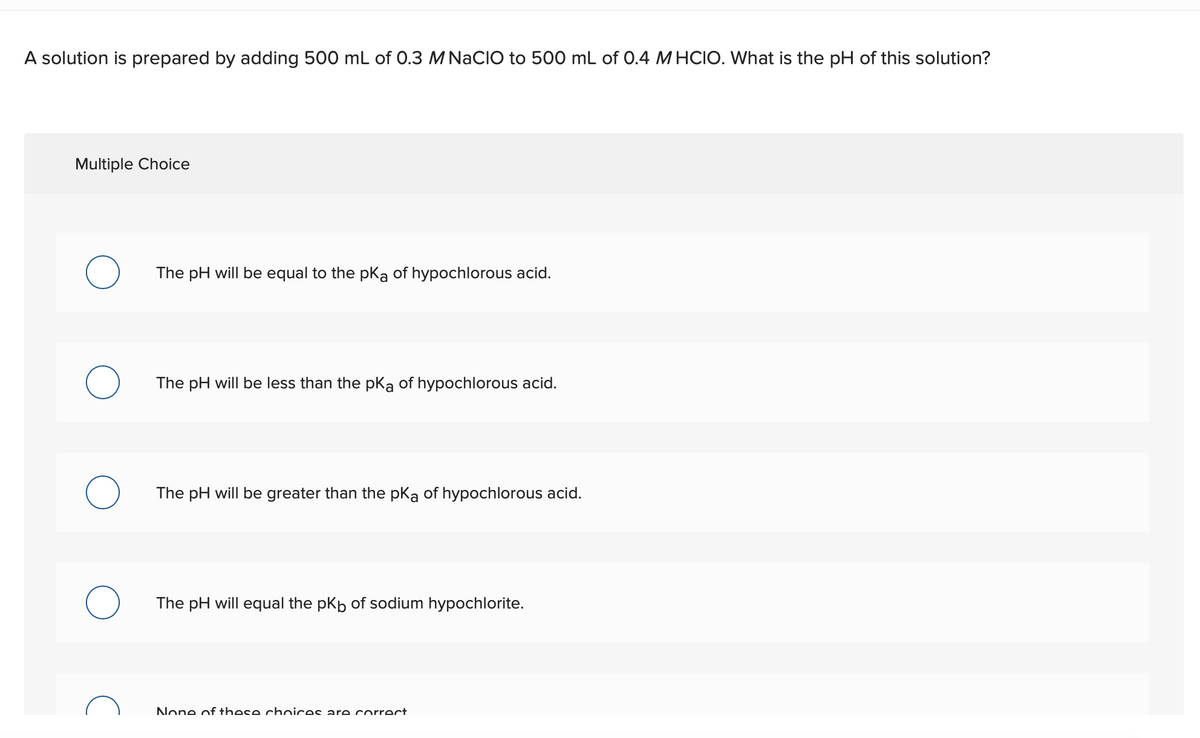
Transcribed Image Text:A solution is prepared by adding 500 mL of 0.3 M NACIO to 500 mL of 0.4 M HCIO. What is the pH of this solution?
Multiple Choice
The pH will be equal to the pka of hypochlorous acid.
The pH will be less than the pKa of hypochlorous acid.
The pH will be greater than the pka of hypochlorous acid.
The pH will equal the pkp of sodium hypochlorite.
None of these chnices are correct
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning