A solution of potassium chromate of unknown concentration was analyzed against a standard of various concentrations. What is the linear equation of the standard curve from the given data? Table 1. Data for Standard Calibration Curve Potassium chromate 4 Absorbance at 425 nm Standard (mol/Liter) 0.00040 0.100 0.00080 0.301 0.00120 0.478 0.00160 0.787 0.00200 0.989 O y = 431.59x +0.9899 O y = 0.9919x + 0.731 O y = 566x -0.1482 Oy=566x +0.1482 none of the above
A solution of potassium chromate of unknown concentration was analyzed against a standard of various concentrations. What is the linear equation of the standard curve from the given data? Table 1. Data for Standard Calibration Curve Potassium chromate 4 Absorbance at 425 nm Standard (mol/Liter) 0.00040 0.100 0.00080 0.301 0.00120 0.478 0.00160 0.787 0.00200 0.989 O y = 431.59x +0.9899 O y = 0.9919x + 0.731 O y = 566x -0.1482 Oy=566x +0.1482 none of the above
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
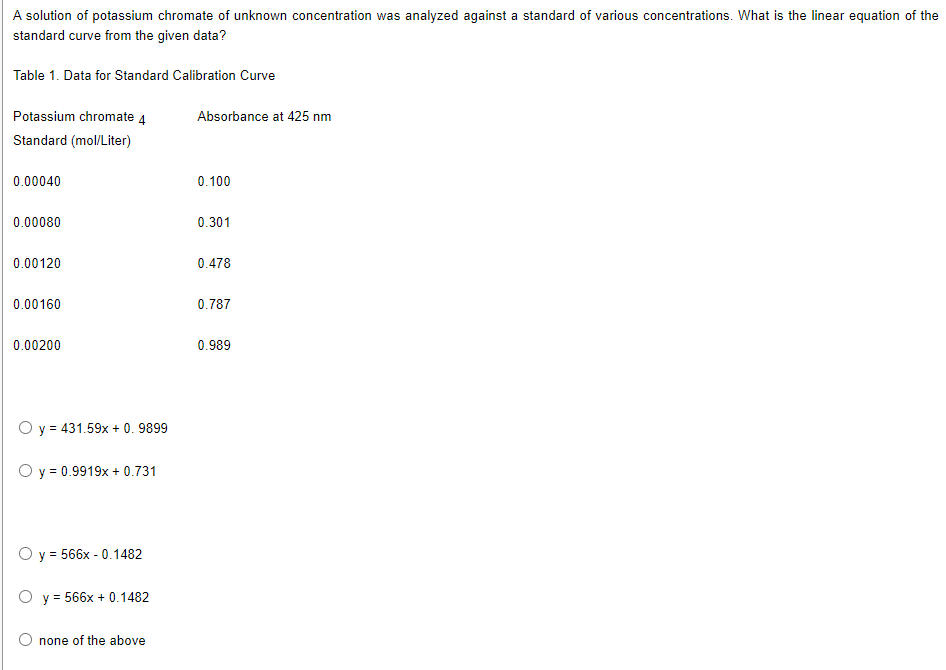
Transcribed Image Text:A solution of potassium chromate of unknown concentration was analyzed against a standard of various concentrations. What is the linear equation of the
standard curve from the given data?
Table 1. Data for Standard Calibration Curve
Potassium chromate 4
Absorbance at 425 nm
Standard (mol/Liter)
0.00040
0.100
0.00080
0.301
0.00120
0.478
0.00160
0.787
0.00200
0.989
O y = 431.59x +0.9899
O y = 0.9919x + 0.731
O y = 566x -0.1482
Oy = 566x + 0.1482
none of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

