B. QUALITATIVE ANALYSIS OF ORGANIC COMPOUNDS Results:(+) or(-) Element Test Name Principal Reagent Chemical Bon/s C. Explain in 5 sentences IN BULLET FORM the ff techniques of separation and give a picture of each separation 1. Chromatography 2. Extraction 3. Recrystallization
B. QUALITATIVE ANALYSIS OF ORGANIC COMPOUNDS Results:(+) or(-) Element Test Name Principal Reagent Chemical Bon/s C. Explain in 5 sentences IN BULLET FORM the ff techniques of separation and give a picture of each separation 1. Chromatography 2. Extraction 3. Recrystallization
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
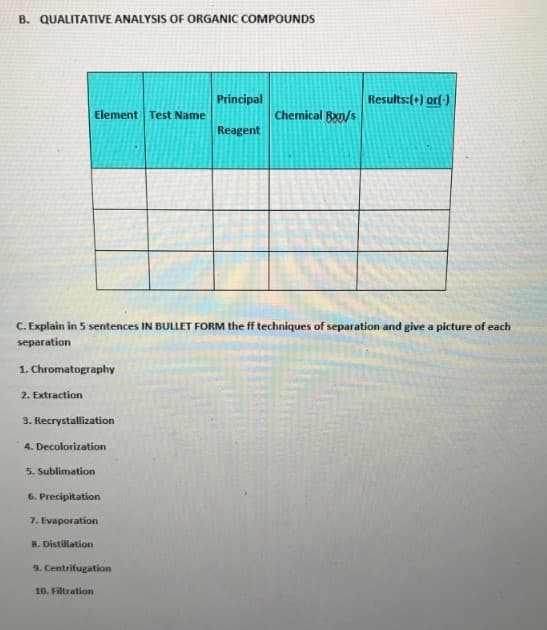
Transcribed Image Text:B. QUALITATIVE ANALYSIS OF ORGANIC COMPOUNDS
Results: (+) or(-)
Element Test Name
Principal
Reagent
Chemical Bxn/s
C. Explain in 5 sentences IN BULLET FORM the ff techniques of separation and give a picture of each
separation
1. Chromatography
2. Extraction
3. Recrystallization
4. Decolorization
5. Sublimation
6. Precipitation
7. Evaporation
8. Distillation
9. Centrifugation
10. Filtration
Transcribed Image Text:References for A & B Experiments
Letter A
A. Ignition test
a. Flammability:https://www.youtube.com/watch?v=43n9-NDrLSE
b. Charring: https://www.youtube.com/watch?v=XcblxxLrj5w
B. Solubility Test
a. & b. https://www.youtube.com/watch?v=LKTe5T00Xdw
c. Urea in water: https://www.youtube.com/watch?v=XNkINFq 3CA
C. Electrical Conductivity: (DO NOT PERFORM THIS AT YOUR HOME!)
Video link: https://www.youtube.com/watch?v=RsGBcP4alDo
Letter B
A. Detection of Carbon And Hydrogen:
https://www.youtube.com/watch?v=xBxXkabvCYU
B. Detection of Elements: Lassaigne's Test:
https://www.youtube.com/watch?v-FUo428gukto
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

