a solution that is 0.115 M in HC10 and 0.170 M in KCIO (K, (HCIO) = 2.9 x 10-8) %3D Express your answer to two decimal places. ΑΣΦ pH %3D a solution that contains 1.07 % C2H;NH2 by mass and 1.19% C2 Hs NH3B by mass (K(C,H;NH2) = 5.6 x 10-4) Express your answer to two decimal places. pH = a solution that has 13.5 g of HC2 H3O2 and 13.5 g of NaC2H3O2 in 150.0 mL of solution (K.(HC2H3O2) = 1.8 x 10-5) Express your answer to two decimal places. ΑΣΦ pH = %3D
a solution that is 0.115 M in HC10 and 0.170 M in KCIO (K, (HCIO) = 2.9 x 10-8) %3D Express your answer to two decimal places. ΑΣΦ pH %3D a solution that contains 1.07 % C2H;NH2 by mass and 1.19% C2 Hs NH3B by mass (K(C,H;NH2) = 5.6 x 10-4) Express your answer to two decimal places. pH = a solution that has 13.5 g of HC2 H3O2 and 13.5 g of NaC2H3O2 in 150.0 mL of solution (K.(HC2H3O2) = 1.8 x 10-5) Express your answer to two decimal places. ΑΣΦ pH = %3D
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter6: Solutions And Colloids
Section: Chapter Questions
Problem 6.71P: 6-71 A 4 M acetic acid (CH3COOH) solution lowers the freezing point by-8°C; a 4 M KF solution yields...
Related questions
Question
Please give me solution part a,b,c properly. Please please
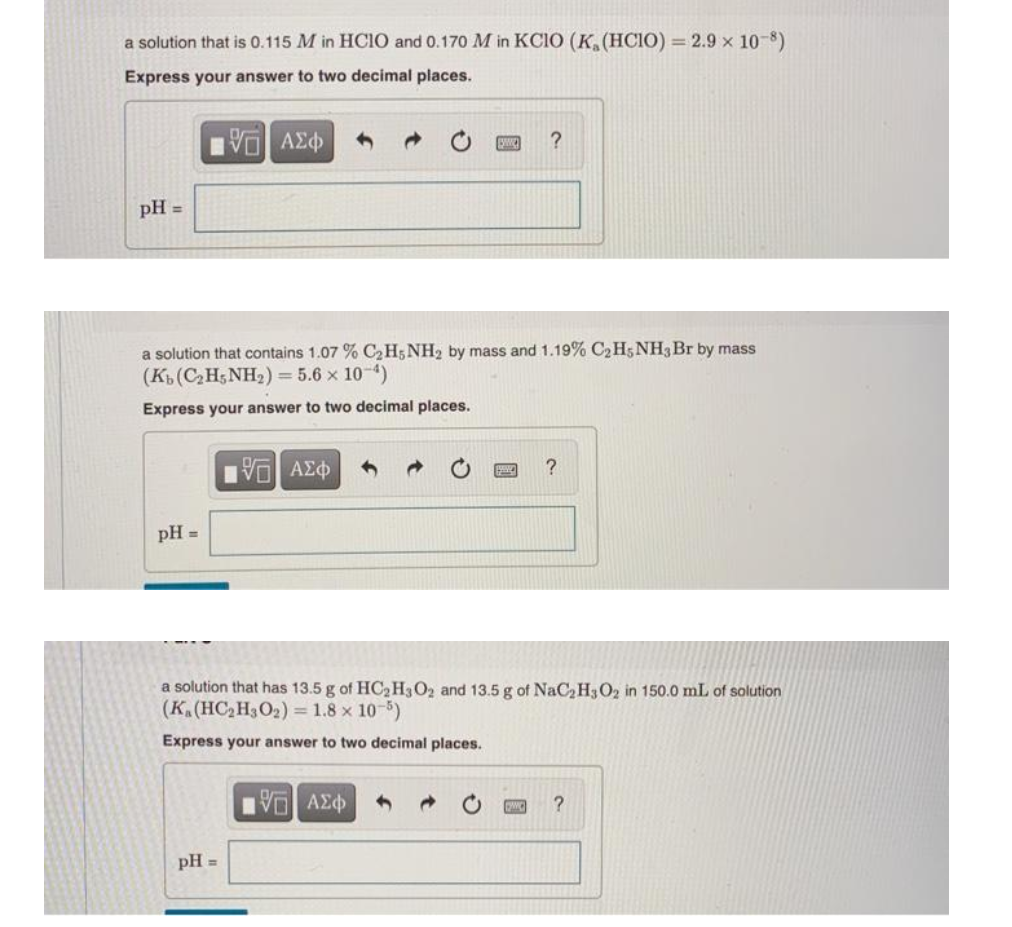
Transcribed Image Text:a solution that is 0.115 M in HCIO and 0.170 M in KCIO (K, (HCIO) = 2.9 x 10-8)
Express your answer to two decimal places.
pH =
a solution that contains 1.07 % C2H5 NH2 by mass and 1.19% C2H5NH3Br by mass
(K(CH;NH2) = 5.6 x 10-4)
Express your answer to two decimal places.
pH =
a solution that has 13.5 g of HC2H3O2 and 13.5 g of NaC2H302 in 150.0 mL of solution
(K.(HC2H3O2) = 1.8 x 10-5)
Express your answer to two decimal places.
pH
%3D
Expert Solution
Trending now
This is a popular solution!
Step by step
Solved in 10 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning
