A standard of solution was put through appropriate dilutions to give the concentrations of glucose (mM) shown in the accompanying table. The absorbances in the table (1.00-cm cells) were recorded at 490 m table. Use the method of least squares to find an equation relating absorbance and the concentration of glucose.
A standard of solution was put through appropriate dilutions to give the concentrations of glucose (mM) shown in the accompanying table. The absorbances in the table (1.00-cm cells) were recorded at 490 m table. Use the method of least squares to find an equation relating absorbance and the concentration of glucose.
Chemistry In Focus
6th Edition
ISBN:9781305084476
Author:Tro, Nivaldo J., Neu, Don.
Publisher:Tro, Nivaldo J., Neu, Don.
Chapter18: The Chemistry Of Food
Section: Chapter Questions
Problem 51E
Related questions
Question
A standard of solution was put through appropriate dilutions to give the concentrations of glucose (mM) shown in the accompanying table. The absorbances in the table (1.00-cm cells) were recorded at 490 m table. Use the method of least squares to find an equation relating absorbance and the concentration of glucose.
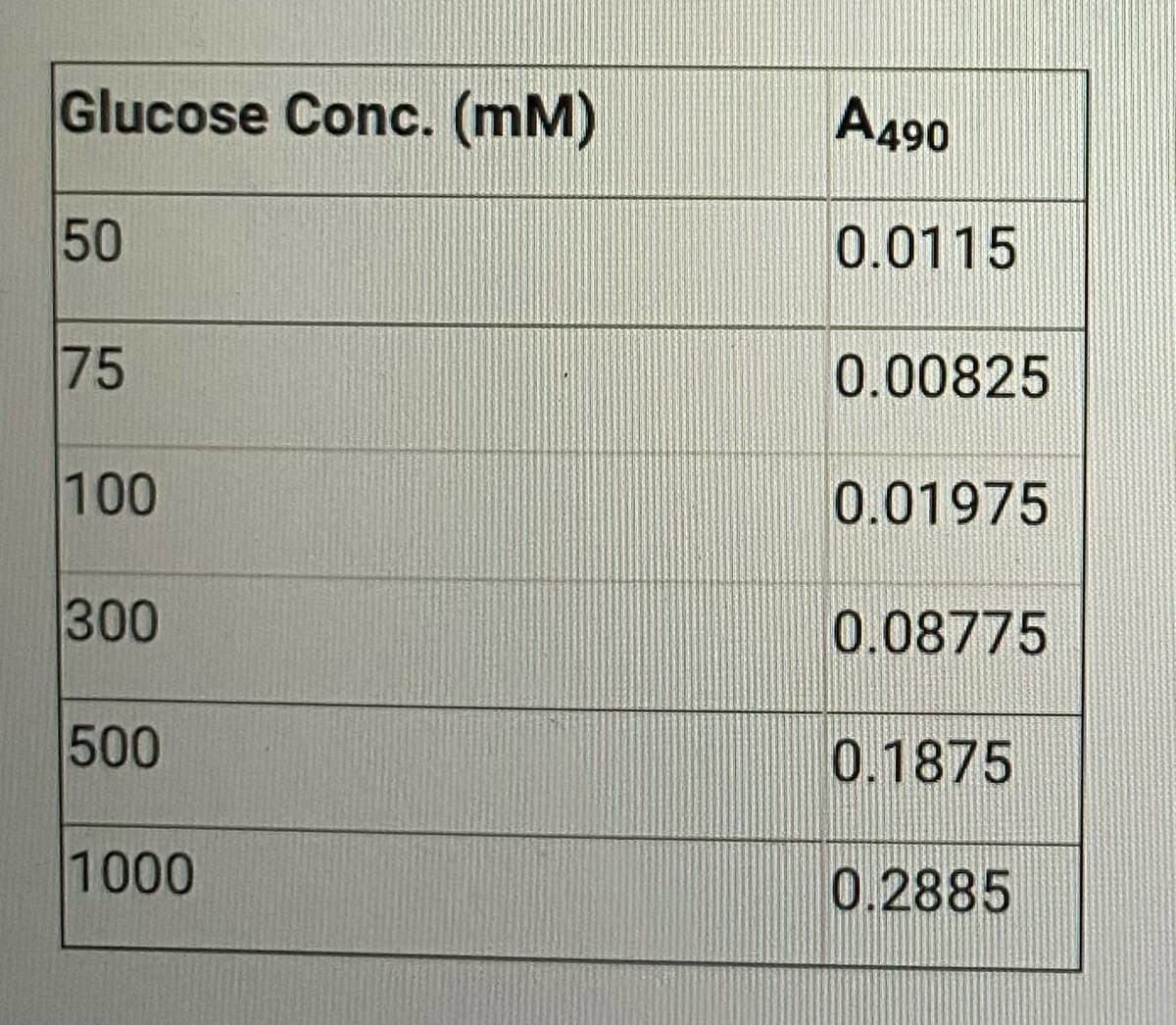
Transcribed Image Text:Glucose Conc. (mm)
50
75
100
300
500
1000
A490
0.0115
0.00825
0.01975
0.08775
0.1875
0.2885
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 14 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry In Focus
Chemistry
ISBN:
9781305084476
Author:
Tro, Nivaldo J., Neu, Don.
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry In Focus
Chemistry
ISBN:
9781305084476
Author:
Tro, Nivaldo J., Neu, Don.
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning