A triprotic acid is titrated with KOH. What species are NOT in solution to any large extent between the 1st and 2nd equivalence points? Select all that apply. O H3A O HA2- A3-
A triprotic acid is titrated with KOH. What species are NOT in solution to any large extent between the 1st and 2nd equivalence points? Select all that apply. O H3A O HA2- A3-
Chapter14: Principles Of Neutralization Titrations
Section: Chapter Questions
Problem 14.38QAP
Related questions
Question
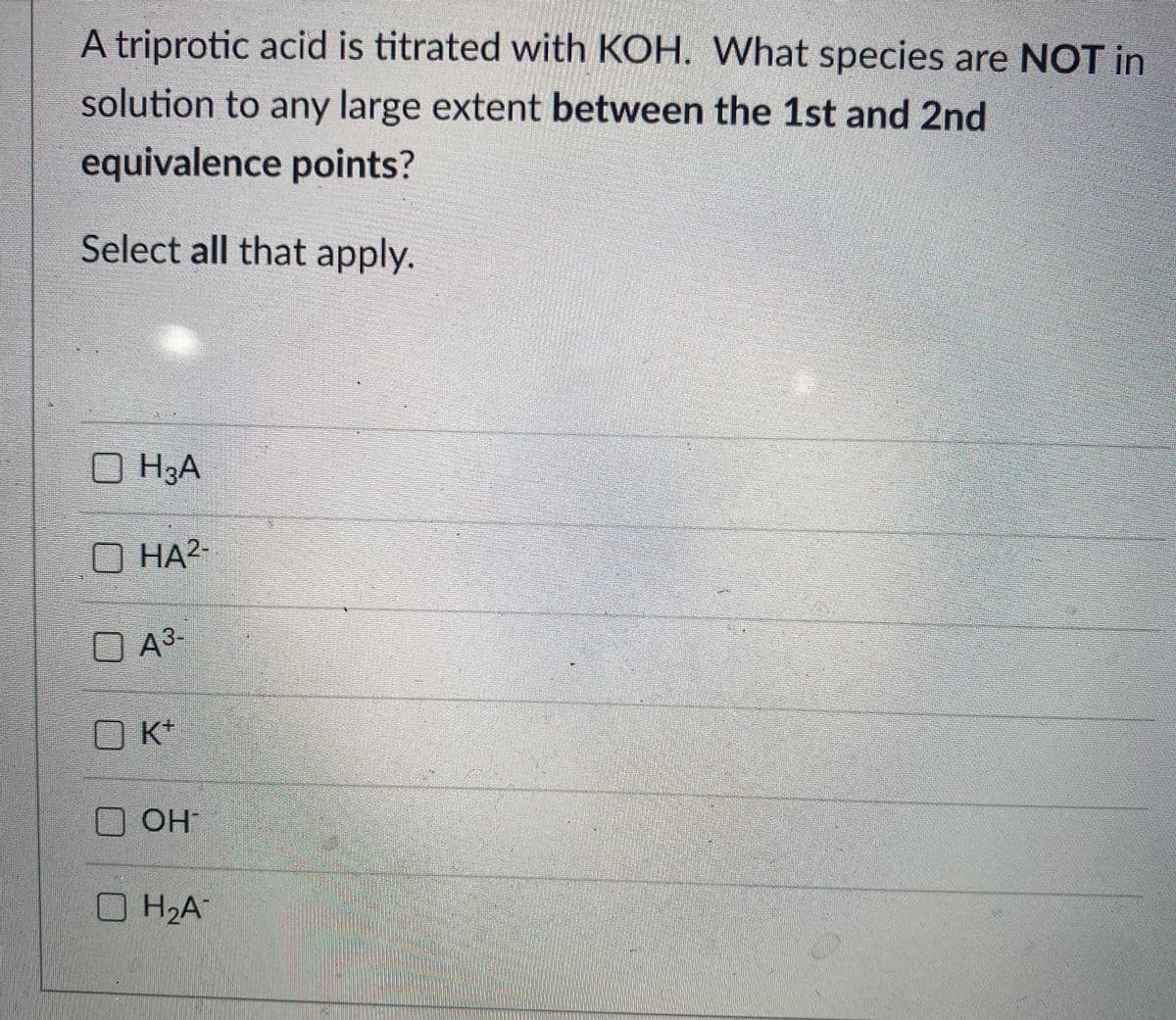
Transcribed Image Text:A triprotic acid is titrated with KOH. What species are NOT in
solution to any large extent between the 1st and 2nd
equivalence points?
Select all that apply.
O H3A
O HA?
O A³-
K+
O OH
H2A
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning