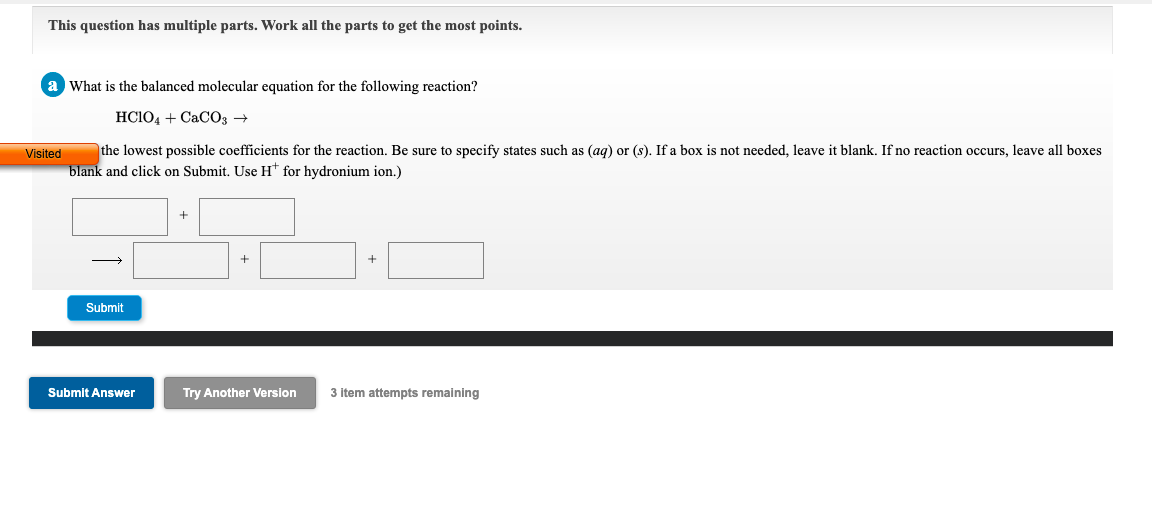
a What is the balanced molecular equation for the following reaction? HCIO4 + CaC03 → ited the lowest possible coefficients for the reaction. Be sure to specify states such as (aq) or (s). If a box is not needed, leave it blank. If no reaction occurs, leave all boxes blank and click on Submit. Use H* for hydronium ion.)
Q: Consider the reaction below: Pd(NO3)4 (aq) + 4 NaCl(aq) PdCl4(s) + 4 NaNO3(aq) ----> The sum of the…
A: To write an ionic equation, we have to write aqueous species as ions and solid species does not…
Q: Write the balanced NET ionic equation for the reaction when Pb(NO3)2 and Kl are mixed in aqueous…
A: The Reaction between Lead Nitrate and Potassium iodide. Pb(NO3)2(aq)+KI(aq) ---------->…
Q: Balance the following reaction. Use the lowest possible numbers. Be sure to put a number in every…
A: To solve this problem we have to balance the given chemical equation .
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A:
Q: (1) Write the net ionic equation for the reaction that occurs when equal volumes of 0.058 M aqueous…
A:
Q: Write the following on a separate sheet of paper, then upload the picture in the Assignment in…
A: Since you have posted a question with multiple sub-parts , we will solve first three subparts for…
Q: A chemical equations representing the reaction of water (HOH) and ammonia (NH3) is shown below.…
A: According to this theory proton (H+) donar is acid and proton acceptor is base acid and its…
Q: 3. Methane (CH) or natural gas is a colorless, odorless gas. Chemical companies add a chemical that…
A: Q.3: Methane (CH4) is a highly reactive compound and ignites immediately with a small amount of…
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three subparts…
Q: Write the balanced COMPLETE ionic equation for the reaction when AuBr₃ and NH₄NO₃ are mixed in…
A: The reactants given are, 1) AuBr3 (aq) 2) NH4NO3 (aq)
Q: - Which one of the following is the correct net ionic equation for the reaction that occurs when…
A: Molecular reaction equation between Pb(NO3)2 and KI : Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq…
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A: Oxidation number is the imaginary or fictitious charge developed on an atom if all shared electrons…
Q: Consider the reaction below: 3 MgCrO4(aq) + 2 FeCl3(aq) --> 1 Fe2(CrO4)3(s) + 3 MgCl2(aq) The sum of…
A: To solve this problem we have to write the net ionic equation of the given reaction .
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: It is basically a type of double displacement reaction. It is also a precipitation reaction. Barium…
Q: 3 Zn(C2H3O2)2(aq) + 2 Na3PO4(aq) → 6 NaC2H3O2(aq) + 1 Zn3(PO4)2(s) Give the Net Ionic Equation for…
A: Please have a look on the solution below:
Q: Balance the following reactions: ___ Zn (s) + ___ HCl (aq) à ___ ZnCl2 (aq) +…
A: According to the law of conservation of mass, the quantity of each element does not change in a…
Q: According to the following reaction, how many moles of phosphoric acid will be formed upon the…
A: The balanced chemical equation for the formation of phosphoric acid is given by
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A:
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: 1) The reactants given are a) K2SO4 (aq) b) FeCl3 (aq) We are required to predict whether a…
Q: Complete and balance the molecular equation Al(OH)3(s)+HNO3(aq) -->
A: When Al(OH)3(s) react with HNO3(aq), aqueous solution of Al(NO3)3 and H2O is formed.
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: Complete the balanced molecular chemical equation for the reaction below. If now reaction occurs,…
Q: Write the molecular, ionicequation and net ionic equations for the following aqueous reactions. If n…
A: As per our guidelines we can only solve first three sub-parts of this question. The solution is as…
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: General concepts.
Q: d. H2C,O4 + CaCI, → CaC,0. + HCI e. HCI + Al > AICI, + H2
A:
Q: Predict the reactants of this chemical reaction. That is, fill in the left side of the chemical…
A: The reaction of acid with base forms salt and water. The products of the given reaction are barium…
Q: . Write the molecular and net ionic equations for the reaction of i. Aqueous hydrochloric acid and…
A: Write the balanced molecular equation Split strong electrolytes to their respective ions for writing…
Q: Write balanced molecular and net ionic equations for thereactions of (a) hydrochloric acid with…
A: Molecular reactions: where two or more than two molecules interact and change the molecules due to…
Q: The reaction 3C12 + 6KOH KCIO3 + 5KCI + 3H2O is the: a. combustion O b. acid-base reaction c.…
A: Correct option - (d) - redox reaction When the potassium hydroxide is treated with chlorine gas…
Q: Write a net ionic equation for the reaction that occurs when solid manganese(II) carbonate is…
A: Balanced chemical equation: In a balanced chemical equation, the atoms of each element should remain…
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of sodium hydroxide…
A:
Q: How do you write a net ionic equation for the formation of gas ex Al2(SO4)3(NH4)2SO4 •24 H2O +…
A:
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: Chemical equation is the equation in which the chemical reaction is represented. All the reactant…
Q: Be sure to specify states such as (aq) or (s). If a box is not needed leave it blank. If no reaction…
A: Neutralization reaction: During a neutralization reaction, an acid and a base produce salt and…
Q: Sulfuric acid dissolves aluminum metal according to the following reaction: 2A1(s) + 3H,SO,(aq) →…
A: 2 Al(s) + 3 H2SO4 (aq) --> Al2(SO4)3 (aq)…
Q: te the balanced formula, complete ionic, and net ionic equations for each of the following acid-base…
A:
Q: What is the product of the net ionic equation, given the following balanced molecular equation?…
A:
Q: Write the balanced formula, complete ionic, and net ionic equations for each of the following…
A:
Q: Write the balanced COMPLETE ionic equation for the reaction when aqueous BaCl₂ and aqueous (NH₄)₂SO₄…
A: Write the balanced COMPLETE ionic equation for the reaction when aqueous BaCl₂ and aqueous (NH₄)₂SO₄…
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A:
Q: 3) CIO H,0, → _cI1 4) CIo _Cro a C+3 5) Br2 → _Br _Bro 6) CIO1
A:
Q: 1. Balance the reaction equations below. a) HCl(aq) + _Mg(OH)2(s) – Н-0() + MgCl2(aq) b) _HNO3(aq) +…
A: Part 1 The balanced chemical reaction is shown as,
Q: Write the balanced NET ionic equation for the reaction when BaCl₂ and ZnSO₄ are mixed in aqueous…
A: Net ionic equation represents the ions responsible for the reaction, which has only the ions…
Q: Write the balanced NET ionic equation for the reaction when BaCl2 and ZNSO4 are mixed in aqueous…
A:
Q: 2. Complete the following equations for reactions of hydrocarbons. a. C̟H10 + O2 b. CH,-CH=CH, +…
A: In this question, we have to complete the following equation for the reaction of hydrocarbons.
Q: box is not needed leave it blank. o reaction occurs leave all boxes blank and click on "Submit". e a…
A: NaOH is a base while nitric acid is an acid. Both are strong base and strong acid and together…
Q: Write the balanced COMPLETE ionic equation for the reaction when aqueous BaCl2 and aqueous (NH4)2SO4…
A: First we will wrote balanced molecular equation, then write ionic equation.
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: In Balanced chemical equation number of atoms of each species are equal on both reactants and…
Q: Mg(OH)2 + H3PO4 – Mg3(PO4)2 + H2O a. Write the balanced chemical equation for the reaction b. Write…
A:
Q: 3. Complete and balance the following single displacement equations. If no reaction takes place,…
A: We have given incomplete single displacement reaction.We need to tell if the reaction is possible or…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- This is my second time submitting this question, please help write out equations even if it is just for the 1st sample so I can apply the values for 2 and 3. Please help me understand the equations for this table. How do I calculate the moles of NaOH and acetic acid? How do I calculate the mass of acetic acid? How do I calculate the molarity and % mass of acetic acid. Thanks! moles of NaOH = 1.0 M x 6.2 mL x 1 L/1,000mL = 0.0062 moles NaOH - from lab manual the experimental data is in the picture with blue answers, the second part of the table I need help with.--------indicates right arrow - The ^ indicates the subscript of element/compound Match these items with type of chemical reaction shown below them? I have indicated below what my answers are – could you verify? ___E__ HNO^3 + CsOH ------- CsNO^3 + H^20 ___A__ H^2 + Ci^2 ------ 2HCI ___B__ NH^4OH ------- NH*3 + H^20 ___C__ 2K + Na^2CO^2 -------- K^2CO^3 + 2Na ___D__ K^2SO^4 + 2NaCI -------- 2KCI + NaSO^4 A – Synthesis reaction B – Decomposition reaction C – Single substitution reaction D – Double substitution reaction E – Neutralization reaction Thank you in advance for your verifcation on types of chemical reactions - I think I have answered correctly, but am just seeking verification.--------indicates right arrow - The ^ indicates the subscript of element/compound Match these items with type of chemical reaction shown below them? I have indicated below what my answers are – could you verify? ___E__ HNO^3 + CsOH ------- CsNO^3 + H^20 ___A__ H^2 + Ci^2 ------ 2HCI ___B__ NH^4OH ------- NH*3 + H^20 ___C__ 2K + Na^2CO^2 -------- K^2CO^3 + 2Na ___D__ K^2SO^4 + 2NaCI -------- 2KCI + NaSO^4 A – Synthesis reaction B – Decomposition reaction C – Single substitution reaction D – Double substitution reaction E – Neutralization reaction Thank you in advance for your assistance.
- For 14 reactions from this experiment in which there is a driving force, give the balanced molecular, ionic and net ionic equations. Then identify the driving force for each reaction. Be sure to include the proper state symbols (e.g., aq, s, I, or g) for all substances. For each type of driving force, be sure to include at least one example (e.g., precipitation, gas formation, neutralization).a. Balance the equation given below. Li2S2O3 +Br2 - LiBr +LiS4O6 B. Write the product of each of the following reactions (remember to add phase labels for each compound) I. Ca(OH)2(ag) +Na2CO3(Ag) - A +B II. C4H10 (g) +O2-C+D C. Write the nett ionic equation for the given below (show all steps) AgNO3(Ag) +NH4I(Ag) - AGI(s) +NH4NO3(Ag)Consider the chemical reaction that takes place between aqueous iron(III) nitrate and aqueous sodium hydroxide. a.) Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs, write NR after the reaction arrow. Be sure to include the proper phases for all species within the reaction. Fe(NO₃)₃(aq) + NaOH(aq) → b.) Write the balanced COMPLETE ionic equation for the reaction. If no precipitation reaction occurs, simply write only NR. Be sure to include the proper phases for all species within the reaction. c.) Write the balanced NET ionic equation for the reaction. If no reaction occurs, simply write only NR. Be sure to include the proper phases for all species within the reaction.
- Complete and balance the equations of the following reactions, each of which could be used to remove hydrogen sulfide from natural gas: 06 (Please Use Cam Scanner or screenshot so that I will not be confuse of subscipt) Appreciate a lot, would give a like after. Thanks! (a) Ca(OH)2(s) + H2S(g) -----> (b) NazCO3(aq) + H2S (g)----->Choose whether each statement below is true or false for the reaction between a solution of HNO2and a solid Ca(OH)2. These reactants can not produce an acid-base reaction because the weak acid is not strong enough to dissolve the solid Ca(OH)2. Assuming this reaction does take place if you write the balanced molecular reaction, in standard form, the reaction shows that for every one mole of the base used in the reaction there are twomoles of water produced. Assuming this reaction does take place if you write the NET ionic reaction contains only one product. HNO2 is a weak acid and therefore cannot react with bases since it does not dissociate in solution.Balance the reaction below. What is the sum of all the coefficients? HIO3+FeI2+HCl = FeCl3+ICl+H2O
- 1) a balanced chemical equation (molecular equation), (2) a total ionic equation, and (3) a net ionic equation for the reactions below. Include the appropriate phase for each species. Do your experimental observations match your predictions? (f) Mg(NO3)2(aq) + NaOH(aq) →Balance the following reaction in acidic conditions: I2 + HNO3 → 2 HIO3 + 10 NO2 What are the coefficients of each of the following when the given equation is balanced? (Note: you have to enter 1 if the coefficient is 1). Blank 1. t. I2 + Blank 2. HNO3 → Blank 3 HIO3 + Blank 4. NO2 + Blank 5. H2Obalance the ff reactions : __Al + __HC2H3O2 = __ Al(C2H3O2)3 + _____H2

