a Write the molecular equation for the reaction of solid zinc carbonate and hydrogen bromide in an aqueous solution. (Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is not needed, leave it blank. Use H for hydronium ion.) ZnCO,(s) + 2HB1(aq) ZnBr, (aq) H,O(1) Co,(g) Correct ZnCO3 (s) + 2HB1(aq) CO2 (g) + H2O(1) + ZnBr2 (ag) b What is the net ionic equation for the reaction of solid zinc carbonate and hydrogen bromide in an aqueous solution? (Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is not needed, leave it blank. Use H for hydronium ion.)
a Write the molecular equation for the reaction of solid zinc carbonate and hydrogen bromide in an aqueous solution. (Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is not needed, leave it blank. Use H for hydronium ion.) ZnCO,(s) + 2HB1(aq) ZnBr, (aq) H,O(1) Co,(g) Correct ZnCO3 (s) + 2HB1(aq) CO2 (g) + H2O(1) + ZnBr2 (ag) b What is the net ionic equation for the reaction of solid zinc carbonate and hydrogen bromide in an aqueous solution? (Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is not needed, leave it blank. Use H for hydronium ion.)
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 81GQ: Balance equations for these reactions that occur in aqueous solution, and then classify each as a...
Related questions
Question
find net ionic equation?
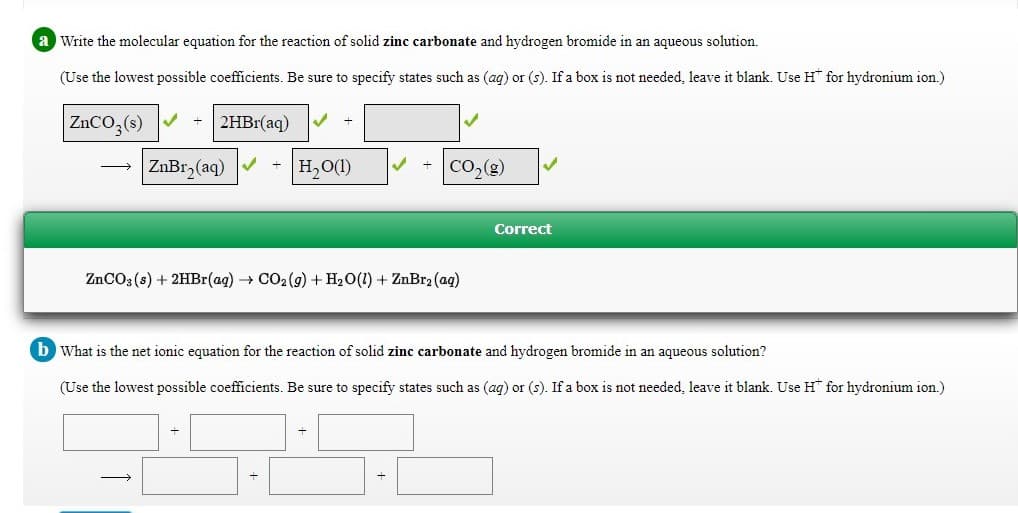
Transcribed Image Text:a Write the molecular equation for the reaction of solid zinc carbonate and hydrogen bromide in an aqueous solution.
(Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is not needed, leave it blank. Use H for hydronium ion.)
ZNCO,(s) V
2HB1(aq)
ZnBr, (aq)
H2O(1)
CO,(g)
Correct
ZNCO3 (s) + 2HBr(ag) CO2 (g) + H20(1) + ZnBr2 (ag)
b What is the net ionic equation for the reaction of solid zinc carbonate and hydrogen bromide in an aqueous solution?
(Use the lowest possible coefficients. Be sure to specify states such as (ag) or (s). If a box is not needed, leave it blank. Use H* for hydronium ion.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax