Balance the following chemical reaction using the combination of a smallest possible set of whole numbers in the blanks. Note: if a reactant(s) and/or product(s) has a coefficient of 1, you must enter a "1" in the appropriate Blank. If you leave the Blank empty, it will be marked wrong. Li2 S(aq) + LİNO3 (aq) + Fe(NO3)3(aq) → Fe2 S3 (s) In Blank #5, classify the reaction type as: A for acid/base, R for redox , and P for precipitation. Blank # 1 Blank # 2 Blank # 3 Blank # 4 Blank # 5
Balance the following chemical reaction using the combination of a smallest possible set of whole numbers in the blanks. Note: if a reactant(s) and/or product(s) has a coefficient of 1, you must enter a "1" in the appropriate Blank. If you leave the Blank empty, it will be marked wrong. Li2 S(aq) + LİNO3 (aq) + Fe(NO3)3(aq) → Fe2 S3 (s) In Blank #5, classify the reaction type as: A for acid/base, R for redox , and P for precipitation. Blank # 1 Blank # 2 Blank # 3 Blank # 4 Blank # 5
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter4: Stoichiometry
Section: Chapter Questions
Problem 4.68PAE: 4.68 The pictures below show a molecular-scale view of a chemical reaction between the compounds AB2...
Related questions
Question
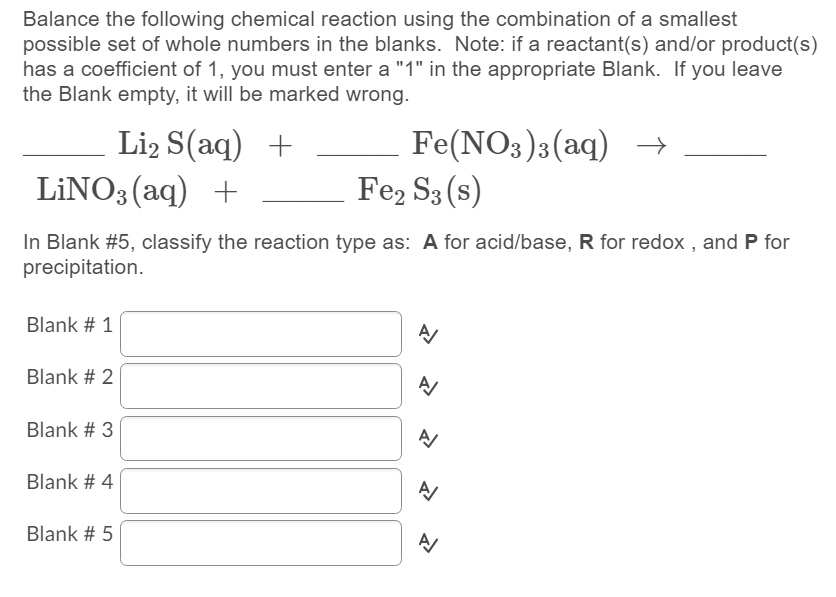
Transcribed Image Text:Balance the following chemical reaction using the combination of a smallest
possible set of whole numbers in the blanks. Note: if a reactant(s) and/or product(s)
has a coefficient of 1, you must enter a "1" in the appropriate Blank. If you leave
the Blank empty, it will be marked wrong.
Li2 S(aq) +
LİNO3 (aq) +
Fe(NO3)3(aq) →
Fe2 S3 (s)
In Blank #5, classify the reaction type as: A for acid/base, R for redox , and P for
precipitation.
Blank # 1
Blank # 2
Blank # 3
Blank # 4
Blank # 5
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning