A. A water soluble sample containing an unknown amount of sulfate ion weighed 0.4892g. When treated according to the procedure of this experiment, the sample produced 0.8462g barium sulfate. What is the mass percent SO42- in the sample?
A. A water soluble sample containing an unknown amount of sulfate ion weighed 0.4892g. When treated according to the procedure of this experiment, the sample produced 0.8462g barium sulfate. What is the mass percent SO42- in the sample?
Chapter88: Column Chromatography
Section: Chapter Questions
Problem 1P
Related questions
Question
Answer A, show all work and use correct number of sig figs.
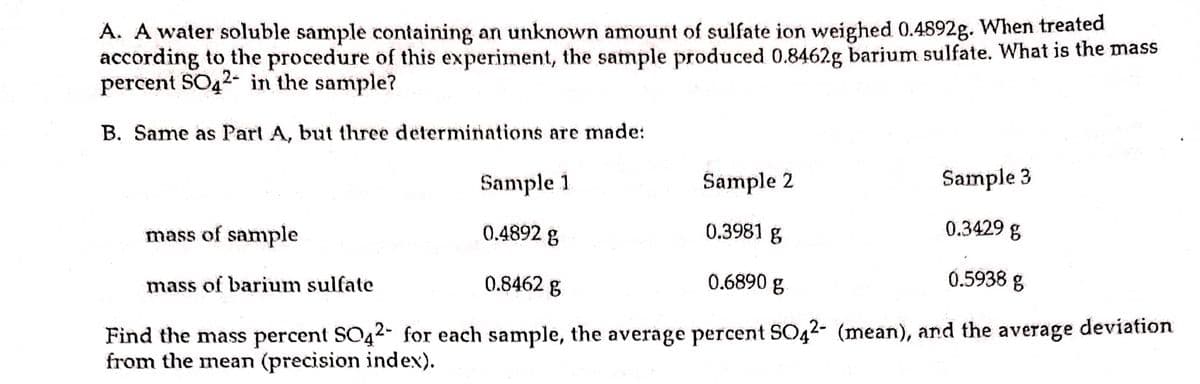
Transcribed Image Text:A. A water soluble sample containing an unknown amount of sulfate ion weighed 0.4892g. When treated
according to the procedure of this experiment, the sample produced 0.8462g barium sulfate. What is the mass
percent SO42- in the sample?
B. Same as Parl A, but three determinations are made:
Sample 1
Sample 2
Sample 3
mass of sample
0.4892 g
0.3981 g
0.3429 g
mass of barium sulfate
0.8462 g
0.6890 g
0.5938 g
Find the mass percent SO42- for each sample, the average percent SO42- (mean), and the average deviation
from the mean (precision index).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT