Q: For the reactionCaCO3(s)CaO(s) + CO2(g)H° = 178.3 kJ and S° = 160.6 J/KThe equilibrium constant for…
A: The objective of the question is to calculate the equilibrium constant for the given reaction at a…
Q: Can you please set up the ice table?
A: The given buffer solution consists of a weak base,C6H5NH2 and its salt ,C6H5NH3Cl.C6H5NH2 is a weak…
Q: Please answer in detail and show all your work, thank you! Synthesis of Ibuprofen-Part 2: Some pain…
A: Synthesis of flurbiprofenStep 1: The ketone starting reactant underwent reductions with sodium…
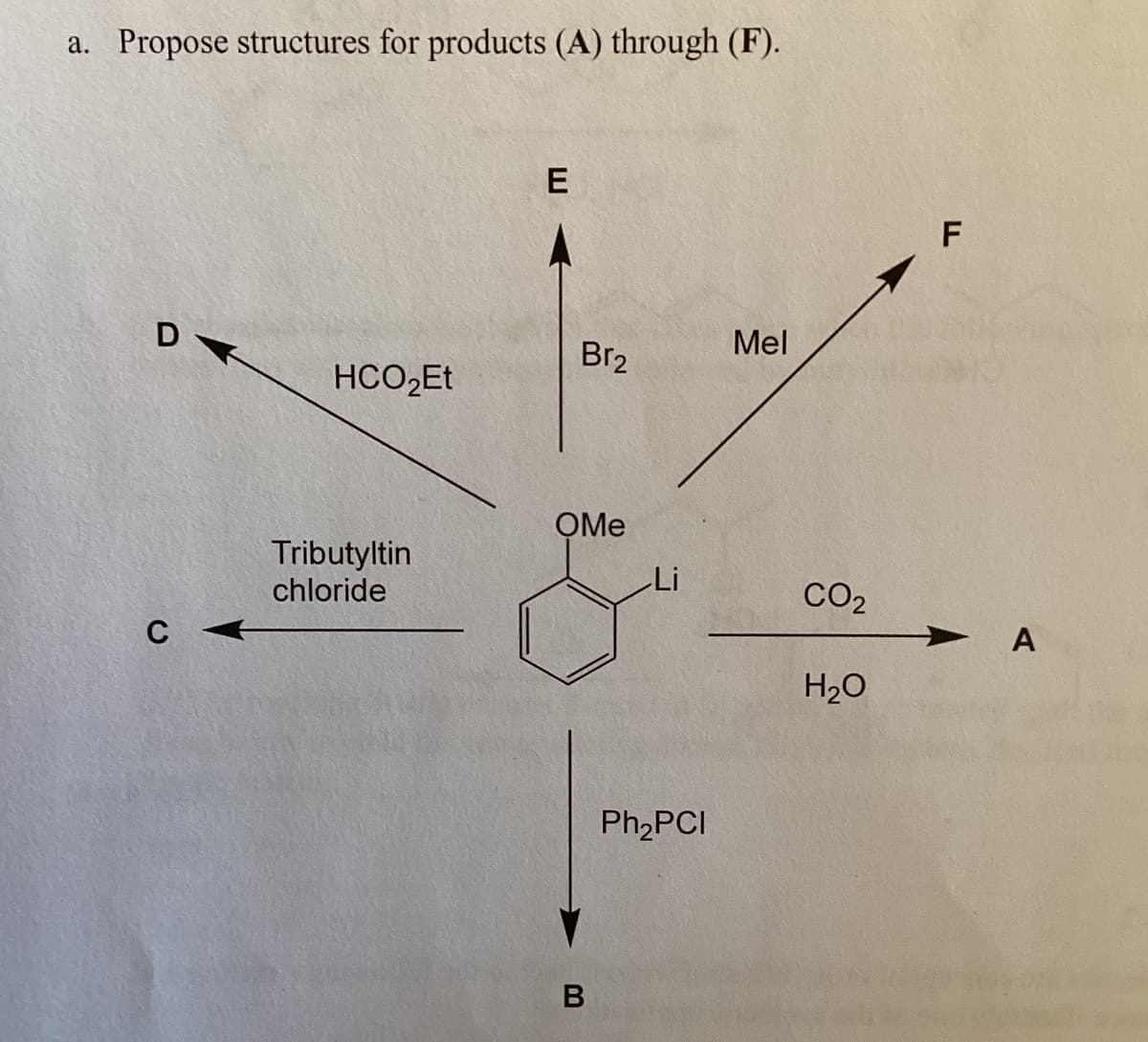
Q: (20 pts) Alcohol A undergoes a remarkable acid-catalyzed conversion to B. Me Me Me HO Me I Me S Me…
A: Step 1: Step 2: Step 3: Step 4:
Q: What is the final temperature when 100.0 g of Al (s = 0.900 J g–1 °C–1) at 0.0°C is placed in…
A:
Q: Pleeesssseeee solve quuuuuuestion 4
A: In the given question we have to determine the empirical formula of benzene, First, we have to…
Q: Please correct answer and don't use hend raiting
A: The molecule on product of the reaction is 2-methylbutanal. It can be synthesized from no more than…
Q: S.m.cem
A: The objective of this question is to provide a comprehensive understanding of dry chemistry…
Q: Using curved arrows, please give a mechanism to explain the following elimination (E1) reaction. H3C…
A: Step 1: Explanation:Alkyl halide, when heated in the presence of ethanol, undergoes elimination…
Q: Huhuhuhuhuu
A: C) The ring with methyl group is activated one because the CH3 group donated electron density inside…
Q: Add curved arrows to show the next step, formation of a peroxy radical by coupling with O2, and…
A: Start by identifying the carbon atoms involved in the reaction. In this case, the 1-bromopropane…
Q: 5) 2 PbS04 → IN PbSO3 + 02 Type 6) _ Al + Fe2O3 → Fe + _Al2O3 5 Type 7) SF6 → S + F2 Typ 51 D 032 8)…
A: Step 1: Step 2: Step 3: Step 4:
Q: Using thermodynamic values from Misra appendix 5, NOT R&H95, write reactions that solubilize…
A:
Q: Use cell potential to calculate an equilibrium constant. Calculate the cell potential and the…
A: E0 Cell = E0 Cathode - E0 Anode = E0 reduction - E0 oxidation = 0.16 - 0.77…
Q: Write the complete equation for and identify the mode of decay for each of the following unstable…
A: The objective of the question is to write the complete decay equation and identify the mode of decay…
Q: Identify the most stable conformation of trans-1-methyl-4-bromocyclohexane (5). A B Me (5) Me Br A…
A: Step 1:
Q: 3. What changes occur to the atomic number and mass of a nucleus during each of the following decay…
A: The symbol of an element is expressed as, A XZ Where, X = the atomic symbol of the element (like…
Q: Calculate quantity of charge and maximum work from a battery. The discharge reaction for a…
A: Step 1:
Q: Part A Account for the difference in the products of the following reactions: ф-оф-о Check all that…
A:
Q: Assuming identical concentrations and temperatures, which of the following weak bases would be…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Choose the best reagents to complete the following reaction.
A:
Q: None
A: The reaction given is a hydroboration-oxidation reaction.In this type of reaction, an -OH group is…
Q: Suggest a structural reason why tertiary alcohols are more reactive than secondary alcohols with…
A: Reactivity with Hydrochloric Acid:Tertiary alcohols are more reactive than secondary alcohols when…
Q: Select the appropriate set of reagents for the below transformation: ? NMe2 A Set A B Set B C) Set C…
A: Let's analyze the reagent sets:Set A (Me₂NPh/Cl⁻):Me₂NPh (dimethylaniline) can act as the…
Q: You want to quickly set up a temporary water bath in your lab with a volume of 10.0 L and a…
A: Step 1:Volume of water is 10LFinal temperature is 37oCInitial temperature of hot water is…
Q: Consider the reaction:CaCO3(s)CaO(s) + CO2(g)Using standard thermodynamic data at 298K, calculate…
A: Step 1:Given reaction:CaCO3(s) → CaO(s) + CO2(g) ΔH0=∑ΔHf0(products)−∑ΔHf0(reactant)…
Q: Use Graph 2 to determine the percentage of parent isotopes that would remain after 2 billion. years…
A: To determine the percentage of parent isotopes remaining after 2 billion years, we can use the data…
Q: According to the equation: Ca(OH)2(aq) + 2HCl(aq) 2H2O (l) + CaCl2 (aq) The balanced ionic equation…
A:
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting and product…
A:
Q: Promethium (Z = 61) is essentially nonexistent in nature as all of its isotopes are radioactive.…
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the phthalic acid…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Step 1:So3(Sulphur trioxide) contains three oxygen atoms and one sulphur atom as central atom. In…
Q: Predict the product CI. Base
A: Alkyl halide reacts with the base and undergoes an elimination reaction, terminal proton is most…
Q: show the mechanism please CH3 NaOH
A: Step 1: OH- is a strong nucleophile and gives SN2 substitution reaction with substrate and…
Q: None
A: Let HA represent the monoprotic weak acid. By definition, a monoprotic weak acid is a weak acid that…
Q: Provide a mechanism and explain the stereochemistry at position 3 and 4 of the product. TIPS OBn 3…
A: The objective of the question is to explain the mechanism of the reaction and the stereochemistry at…
Q: Understanding the optical rotation that we observe in lab is not the same as the specific rotation…
A: here we need to use biot's lawgiven: α= 5.5 l= 1dm c= 0.083 g/ml experimental specific rotation =…
Q: What is an appropriate stepwise synthesis for the following synthesis that uses 1-propanol as the…
A: Step 1: 1-propanol reacts with PCC/CH2Cl2 to form the corresponding aldehyde. Step 2: The aldehyde…
Q: None
A: Step 1:The Diels-Alder reaction is a concerted, pericyclic reaction involving a 4+2 cycloaddition…
Q: A titration was performed using a 25.00 mL sample of 0.15 M weak acid and 0.092 M strong base. What…
A: In this problem, the point of titration is after the equivalence point. Here are some notes to…
Q: Provide a synthesis of the target molecule. All of the carbon atoms in the final product must come…
A: Step 1: Wittig reaction:Methyl bromide with triphenyl phosphine and n-BuLi gives phosphorus…
Q: Draw the arrow-pushing mechanism of the following reaction: OH HO H+ Draw the arrow-pushing…
A:
Q: 2. When an anhydrous solution of CrCl₂ in EtOH was treated with ligand 1 (below), a precipitate of…
A: (a) Analytical Data Interpretation and Possible Structures for X and YGiven the analytical and…
Q: O Chemical Reactions Using molarity to find solute mass and solution volume Bis 1/3 Calculate the…
A: Molarity is a unit of concentration expressed as the number of moles of dissolved solute per liter…
Q: 3 1/1 point Silver bromide has a solubility of 0.133 mg per 1.00 L of water. Find Ksp for silver…
A:
Q: Heating D-altrose with dilute acid produces a nonreducing anhydro sugar (C6H1005). Methylation of…
A: The objective of the question is to understand the structure of the anhydro sugar formed by heating…
Q: What is the pH of a 0.30 M solution of NH3? (Kb 1.8 x 10-5) a) 6.84 b) 8.32 c) 11.43 d) 7.09 e) 7.99…
A: Step 1:
Q: :$:$;);$):):$$:$;$;$
A:
Q: Use the standard reduction potentials shown here to answer the questions. A copper, Cu(s), electrode…
A:
Q: 2. Starting with 2-methyl-1-phenylpropan-1-one, show how you can convert this to 2-methyl-1-…
A: The objective of the question is to convert 2-methyl-1-phenylpropan-1-one to…
Step by step
Solved in 2 steps with 3 images

- Write all the isomers of [PtCl(CO)(PMe3)(C2H4)]+ and use the trans directing effect of ligands to suggest the reaction pathways to synthesize the above isomers from [PtCl4] 2-. Discuss the truth of the following statement. Explain why it is true or false Every SN1 reaction produces racemic mixtures in the productsProvide the names for the following?
- What is the form that Phenazopyridine found , Crystaline or amorphus? And at what range of PH it is Soluble? And is it Soluble in acidic or basic medium?Please provide structures for the given reactions NN, OO, and PPBriefly explain (in one or two sentences) the following observations:a. Unlike NH3, BiH3 is very unstable compound.b. Solid SO3 forms a trimer (SO3)3 but solid SeO3 forms a tetramer (SeO3)4.c. NH3 can be obtained from N2 and H2, but PH3 has to be prepared from PCl3 and LiAlH4
- Provide an explanation for this order of reactivity.a) Answer the following questions regarding solvent polarity and reaction rates.i. Suggest how to increase the rate of reaction if ions are produced as the transition state. Give your reason ii. What would be the effect of polarity of solvents towards a reaction which produces a neutral substance as the transition state?Briefly explain about Degradation of azo dyes , include aerobic, anaerobic etc.

