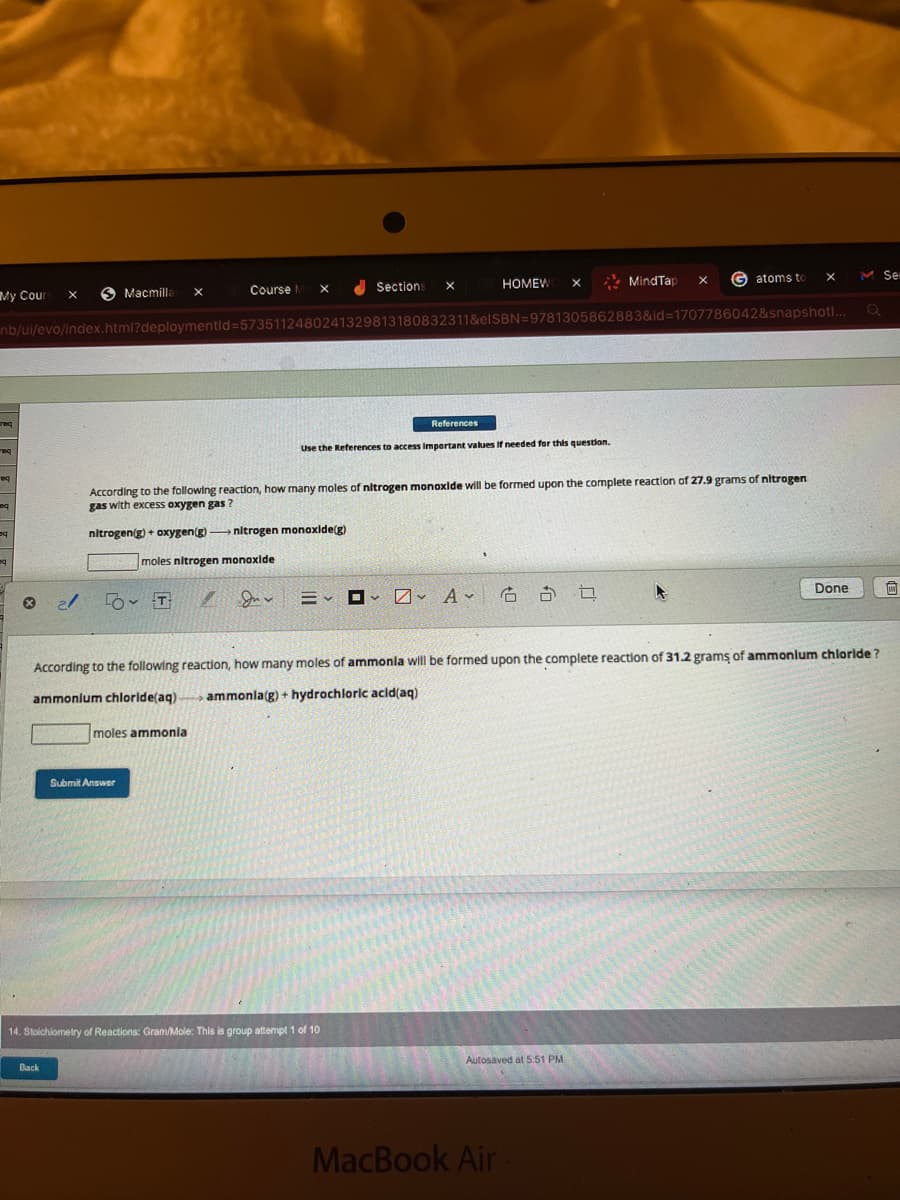
According to the following reaction, how many moles of nitrogen monoxide will be formed upon the complete reaction of 27.9 grams of nitrogen gas with excess oxygen gas? nitrogen(g) + oxygen(g) - →→nitrogen monoxide(g)
According to the following reaction, how many moles of nitrogen monoxide will be formed upon the complete reaction of 27.9 grams of nitrogen gas with excess oxygen gas? nitrogen(g) + oxygen(g) - →→nitrogen monoxide(g)
Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
Transcribed Image Text:My Cour
req
BQ
eq
q
q
eq
X
Macmilla X
nb/ui/evo/index.html?deploymentid=5735112480241329813180832311&elSBN=9781305862883&id=1707786042&snapshot...
Back
2/ OT
Course M
X
moles ammonia
Submit Answer
Sections
X
References
14. Stoichiometry of Reactions: Gram/Mole: This is group attempt 1 of 10
HOMEW X
Use the References to access Important values if needed for this question.
According to the following reaction, how many moles of nitrogen monoxide will be formed upon the complete reaction of 27.9 grams of nitrogen
gas with excess oxygen gas?
nitrogen(g) + oxygen(g) →→→→nitrogen monoxide(g)
moles nitrogen monoxide
J = □ □ A 6 q
MindTap
X
Autosaved at 5:51 PM
MacBook Air
Gatoms to
According to the following reaction, how many moles of ammonia will be formed upon the complete reaction of 31.2 grams of ammonium chloride?
ammonium chloride(aq) ammonla(g) + hydrochloric acid(aq)
X
Done
M Ser
Q
M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



