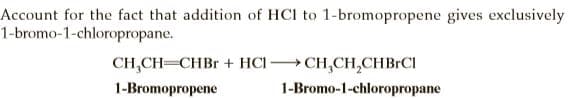
Account for the fact that addition of HCI to 1-bromopropene gives exclusively 1-bromo-1-chloropropane. CH,CH=CHBR + HCI CH,CH,CHBRCI 1-Bromopropene 1-Bromo-1-chloropropane
Q: ) 3-methyl_l-butene H2O/ IHB ) 4-methy lpenta -1,3-diene cl2/ccly.
A: The question is based on concept of organic reactions. we have been given reactant. We have to…
Q: Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: 1. Classify the following hydrocarbons as: Cycloalkane Cycloalkene Cycloalkyne Alkanes Aromatic…
A:
Q: The heat of hydrogenation of cis-2,2,5,5-tetramethyl-3-hexene is -154 kJ (-36.7 kcal)/ mol, while…
A:
Q: Illustrating with equations, indicate how the following substrates or reactants would be converted…
A: A chemical reaction in which one group is replaced by another group is called substitution reaction.…
Q: b) Name the following molecule, excluding stereochemistry OH Cl e-chloro-1- bhe
A: Since you have asked multiple question in the same request, we will solve the first question for…
Q: A hydrocarbon of molecular mass 72 g mol–1 gives a single monochloro derivative and two dichloro…
A: Different organic compounds can have different molecular structures due to bonding of different…
Q: Please do the following . 2-butyne + HgSO4, H2SO4 and H2O 2-methyl-1,3-butadiene + HC ≡ C –…
A: Note: Since you have posted multiple independent questions in the same request ,we will solve the…
Q: Identify the SYSTEMATIC name of this aliphatic hydrocarbon * 1-pentenylcyclopentane O…
A:
Q: When bromo cyclo-butane interacts with Mg then H20 , it gives cyclo-butane IO butene .II O…
A: The question is based on the concept of organic reactions. We have to identify the product formed.…
Q: (3E,5Z)-4-methyl-3,5-nonadiene (4E,6Z)-4-(1-methylethyl)-6-methyldodecadiene
A: According to the IUPAC rule, 1. Identify longest carbon chain then numbering of main chain from that…
Q: Name the alkene. Be sure to indicate stereochemistry and use hyphens (-) not endashes (-). H3C-CH2…
A: First numbering the longest chain possible. Then numbering in such a way that alkene group gets…
Q: Which among the following is a terminal alkene? propene cyclohexene cis-2-Butene 1-butyne
A:
Q: Bromine adds to cis- and trans-2-butene to give different diastereomers of 2,3-dibromobutane. What…
A: According to the question, we get that bromine adds to cis- and trans-2-butene to give different…
Q: Draw structural formulas of all chloroalkanes that undergo dehydrohalogenation when treated with KOH…
A:
Q: Give the structure corresponding to following name ? (1R,2R)-trans-1-bromo-2-chlorocyclohexane
A: Introduction: We have to draw the structure of (1R,2R)-trans-1-bromo-2-chlorocyclohexane.
Q: Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination…
A: Isolated diene These are the compounds in which there are more than one single bond is present…
Q: Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination…
A: Isolated diene: Those diene in which there is separation between double bonds due to two or more…
Q: From : cis (1s, 4s) -1-bromo- (tert-butyl) cyclohexane trans (1r, 4r)- 1- iodo- 4-…
A: Markovnikov addition is used to predict the stereochemistry and regiochemistry of unsymmetrical…
Q: Some of the following examples can show geometric isomerism, and some cannot. Forthe ones that can,…
A: Usually for a compound to exhibit geometrical isomerism, a restricted rotational bond is necessary…
Q: from the addition of one mole of HBr to one mole of Buten-1 is obtained: a) Bromo-1-butane b)…
A: The regioselectivity of addition reactions of alkenes and alkynes is predicted by this rule. In an…
Q: 1,4-dimethylcyclohex-1-ene with BH3, H;O2, OH 4-methylpent-2-ene with mCPBA, H;O, H*
A: Given reactions are : And Give the product of the following compounds = ?
Q: Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination…
A: When the double bonds are separated by more than one single bond, that alkene is called isolated…
Q: Draw a structural formula for 3-ethylcyclopentene. • You do not have to consider stereochemistry. •…
A: Drawing structural formula from IUPAC name:- First identify what is word root ( number of carbon…
Q: ) 4-metylpenta-1,3-diene c2/ccly4
A:
Q: What is the IUPAC name for the following molecule? O (2E,5Z)-2-ethyl-5-methylhepta-2,5-diene O…
A: The first and the foremost step while naming the IUPAC name of an organic compound is the…
Q: 10. Cyclohexane (CH12)
A: Simple forms, colours, and textures are used to create three-dimensional drawings. The entity being…
Q: Name the alkene. Be sure to indicate stereochemistry and use hyphens (-) not endashes (-). H2C-CH3…
A:
Q: Which substance is not a structural isomer of hexyne? O hex-2-yne O hex-3-yne O…
A:
Q: Name the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H H H3C CH3 CH;CH2CH2…
A:
Q: Please name the following compound. (3S, 10S,Z)-10-((S)-sec-butyl)-3-methyltetradec-7-ene (3S,…
A: The naming of any compound is done by the number of carbon atoms present in parent chain and the…
Q: 3,4-Dimethylpent-1-ene has the formula CH2“CH¬CH(CH3)¬CH(CH3)2. When pure (R)-3,4-dimethylpent-1-ene…
A: Hydrogen in the presence of platinum is a reducing agent which reduces alkene to alkane. Here in…
Q: Which of the fallowing names are/is incorrect?* (a) 1-Methylcyclopent-2-ene (b) 1-Methylpent-1-ene…
A: We will choose Correct statement.
Q: Prepare the following compounds from methylcyclohexene: a. 1-chloro-1-methylcyclohexane b.…
A:
Q: Write the structural formula for each of the following compounds. a) sec-Butylcycloheptane oniwollot…
A: In the given question, the structural formula of the given compounds is to be determined. The given…
Q: 3. The following alkenes can be prepared by dehydration of an appropriate alcohol. Show the…
A: Hello ! Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the…
Q: Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination…
A: Diene is a covalent compound containing two double bonds. Isolated diene is that having double bond…
Q: re is the chemical structure of 1-chlore-2-methypropane HH H H-C-C-c-ci: H H-C-H H. ide whether each…
A: If the molecules have different numbers of C atoms , then these molecules are completely different…
Q: Br Br CH3 H3C. Br H3C. CH3 Br Br H3C CH3 Br Br CH3 H3C.
A:
Q: The name of this compound. Br H. C=C Cl F O Z-2-bromo-2-chloro-1-fluoroethene…
A: Rules for naming a compound :
Q: Name the alkene. Be sure to indicate stereochemistry and use hyphens (-) not endashes (- H2C-CH3…
A: Name of the given hydrocarbon can be given according to IUPAC Nomenclature rules.
Q: (Specify (E)(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to…
A: IUPAC naming in cyclic organic compounds.
Q: The correct IUPC name of the following compound is A. (Z)-4-ethyl-2-methylhexa-1,4-diene B.…
A: Select the longest carbon chain. Numbering Naming (prefix + word root + suffix)
Q: Predict the products of the following reactions. cyclopentadiene + methyl acrylate, CH2“CH¬COOCH3
A: The structure of cyclopentadiene is shown below. The structure of methyl acrylate is shown below.…
Q: When bromo cyclo-butane interacts with Mg then H20 , it gives .I суclo- butane .II сyclo- butene…
A:
Q: structures
A:
Q: Which of the following alkenes do not show cis-trans isomerization? I) 2-methyl-2-hexene II)…
A: Since you have posted multiple questions, we are entitled to answer the first only. 1) The compounds…
Q: Classify the following dienes and polyenes as isolated, conjugated, cumulated, or some combination…
A: Isolated dienes are separated by more than one single bond. Conjugated dienes are separated by one…
Step by step
Solved in 2 steps with 1 images

- What’s the role of NaBH4 in Oxymercuration-Demercuration of cyclohexene?Please tell the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. Also please list how many isomers can be formed in each case.A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2 CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under the same conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C. Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structures of A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions.
- You are required to synthesize 2-bromopentane from the reaction between an alkene with HBr. Which alkene, 1-pentene or 2-pentene, should you react with HBr in order to get 2-bromopentane? Give an explanation.Free-radical chlorination of hexane gives very poor yields of 1-chlorohexane, while cyclohexane can be converted to chlorocyclohexane in good yield. How do you account for this difference?Which of the following is most soluble in basic medium? a. cyclopropane b. 1,3-cyclobutadiene c. 1,3-cyclopentadiene d. benzene
- A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactionsReaction of HBr with 3-methylcyclohexene yields a mixture of four products: cis-and trans-1-bromo-3-methylcyclohexane and cis-and trans-1-bromo-2-methylcyclohexane. The analogous reaction of HBr with 3-bromocyclohexene yields trans-1, 2-dibromocyclohexane as the soleproduct. Draw structures of the possible intermediates, and then explain why only a single product is formed in this reaction.From : cis (1s, 4s) -1-bromo- (tert-butyl) cyclohexane trans (1r, 4r)- 1- iodo- 4- isoproppylcyclohexane 1,5 - dimethylcyclohex -1 -ene trans (1r, 4r)- (tert - butyl) - 4- fluorocyclohexane trans (1r, 4r) - 1- bromo - 4- (tert - butyl) cyclohexane 1,3,5 - trimethylcyclohex - 1- ene . 5- Choose the substrate from the ones that drawn and illustrate an example complete Markovnikov addition mechanism using the reagents of your choice showing all stereochemical consequences. Identify if your addition mechanism is syn addition or anti addiction (Use curved arrows to show the electron movement in everyday steps). use ChemDraw if possible..
- Which will be more stable, cis or trans-1,4-tert-butylcyclohexane? Explain by drawing their structures?Give the structure corresponding to following name ? (1R,2R)-trans-1-bromo-2-chlorocyclohexaneThe heat of hydrogenation of cis-2,2,5,5-tetramethyl-3-hexene is -154 kJ (-36.7 kcal)/ mol, while that of the trans isomer is only -113 kJ (-26.9 kcal)/mol. Q.) Why is the heat of hydrogenation of the cis isomer so much larger (more negative) than that of the trans isomer?

