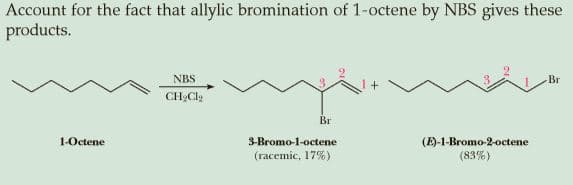
Account for the fact that allylic bromination of 1-octene by NBS gives these products. NBS Br CH,Clg Br 1-Octene 3-Bromo-1-octene (E)-1-Bromo-2-octene (83%) (racemic, 17%)
Q: Elimination occurs when (Z)-3-bromohex-3-ene is treated with NaNH2. Under the same conditions,…
A: The elimination reaction occurs when (Z)-3-bromohex-3-ene is treated with NaNH2 is as shown below.
Q: 33. Arrange each set of compounds in order of reactivity towards the E2 process. Br Br Br CI (a) (b)
A: Interpretation- To arrange set of compounds in the order of reactivity towards E-2 process .…
Q: Classify the following diene, as an aid to understanding its reactivity. H,C=C%3DCH, Conjugated…
A: The three different types of dienes are 1) Conjugated dienes 2) cumulated dienes 3) Isolated…
Q: Provide the reagents necessary to complete the following transformation. Hint: be sure to use the…
A: In this question we have to tell the reagent used for the conversion of the given reaction.
Q: Addition of Br2 to (1)-hex-3-ene produces Wameso dibromide Branisture of enantiomeric dibromides…
A: First the structure of (E)-hex-3-ene is as follows,
Q: a. What is the major monobromination product of the following reaction? Disregard stereoisomers. b.…
A:
Q: Which statement is true? Options: А: Upon photo-excitation y4 is the LUMO of the pentadienyl anion.…
A: When the true statement of the photo-excitation is given below,
Q: 1-ВНСу2, ТHF 2 HаО2, NaOН ОН HO ÓCH3 E LOH
A: We have to carry out the given synthesis. We also give the structures of the reactants, products and…
Q: A hetero Diels-Alder reaction is a variation of the Diels-Alder reaction in which one or more carbon…
A: We have find out reaction mechanism.
Q: What type of sigmatropic rearrangement is illustrated in each reaction?
A: (a) The pi bond is rearranged and the σ bond is broken in this sigmatropic rearrangement. This…
Q: 5) Construct the orbital correlation diagram for hexatriene-cyclohexadiene interconversion, to what…
A: Since you have posted multiple questions, we will answer the first one for you. To get the remaining…
Q: [3] Reaction of either 3-bromo-1-butene or (Z)-1-bromo-2-butene with water under S, 1 condition…
A:
Q: The following questions ask you to choose which of the pair reactions shown has been correctly…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: 1. Predict the products of the following reactions, showing stereochemistry and regiochemistry where…
A: a. In presence of Lindlar's catalyst cis-alkene is formed. b. When excess hydrogen are present then…
Q: How many distinct alkene products are possible when the alkyl bromide below undergoes E2 elimination…
A: In E2 elimination reaction,the proton and the leaving group removed simultaneously by the base and…
Q: KMNO4 6. CH3 + CN- ´Br 7. NaOEt ELOH elimination product 8. Br
A:
Q: но HO.
A:
Q: ON=N=N 1,3 dipolar cycloadditon =ル ONEN=N 1,3 dipolar cycloadditon シ=を
A:
Q: 1) Which of the diene isomers is not suitable for 1,2- and 1,4-addition as well as [4+2]…
A:
Q: What type of sigmatropic rearrangement is illustrated in each reaction?
A:
Q: Predict the major product from the following Dieckmann cyclizations. OCH3 1. косн H,O*, heat ÓCH, 2.…
A:
Q: When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40%…
A: The structure of buta-1,3-diene is shown below.
Q: 3-bromo-1-pentene and 1-bromo-2-pentene undergo SN1 reaction at almost the same rate, but one is a…
A: Generally, alkyl halide or alcohols undergo elimination reaction which give rise to alkene.…
Q: Complete the following reaction scheme. Give all product(s) and indicate major or minor and any…
A: M . Br2 /H2O : At first brominuim ion was formed then anti attack by OH - on more substituted site…
Q: addition of HBr
A:
Q: Rank the following in order from slowest to fastest rate of reaction in a Diels–Alder reaction with…
A: Introduction: Diels-Alder reaction is a cycloaddition reaction of diene and dienophile to form a…
Q: The elimination reaction of butan-2-ol will produce butene and but-2-ene via mechanism O a. SN1 O b.…
A:
Q: Rank the following organobromides in order of increasing rates of ionization to its carbocations and…
A:
Q: 5) A diene is a compound that contains two double bonds. When 1,3-cyclohexadiene reacts with DBr…
A: During the reaction the carbonation is formed which rearranges to form the most stable form and…
Q: 4. For the following reactions, develop a transition-state structure using molecular models which…
A: All the transition states are given in third bracket. 4a) Reaction type: Electrocyclic reaction…
Q: 21. Provide all possible E2 products with the correct stereochemistry. Label the major elimination…
A: Given
Q: LOCH3 СООН „CH3 NO2 H. H. I II III IV
A: Dienes and dienophile react with each other to give diels Alder reaction.
Q: Which is the main product when 2-methylbuta-1,3-diene reacts with one equivalent of HBr at room…
A:
Q: Solve this question
A: SN1 reaction does not lead to inversion in configuration whereas SN2 reaction leads to inversion in…
Q: The aryl diene undergoes sequential Heck reactions to give a product with the molecu- lar formula…
A: Given reaction,
Q: a) What product is formed from the [1,7] sigmatropic rearrangement of a deuterium in the following…
A: (a) The reactant in the equation undergoes [1, 7] sigmatropic rearrangement of a deuterium to…
Q: The following alkenes undergo Diels-Alder reactions EXCEPT: A. В. || D. C. NC || +
A:
Q: HBr (excess) Br Br
A: Firstly, the addition of H+ takes place across the triple bond which gives rise to two carbonations,…
Q: 1. Predict the products of the following reactions, showing stereochemistry and regiochemistry where…
A:
Q: What diene and what dienophile should be used to synthesize the following?
A: Retro-Diels Alder reaction of a Diels-Alder product is an inverse process of Diels-Alder reaction.…
Q: The reaction of butan-2-ol with concentrated aqueous HBr goes with partial racemization, giving more…
A: The answer for first part is given below. Kindly repost the other part as separate one
Q: Draw the alkene products including E,Z isomers that might be obtained by dehydrohalogenation of…
A:
Q: Predict the major products of the following reactions.(a) 1@ethylcycloheptene + ozone, then…
A: Ozonolysis of an alkene results in the breakdown of the double bond and formation of two molecules…
Q: HO ОН
A: The first step of the reaction is the Diels-Alder reaction and the product of the Diels-Alder…
Q: Which alkyl bromide(s) can form the alkene under E2 elimination conditions. Na CH;CH,OH Br Br. Br A…
A:
Q: Predict the major products of the following reactions. Include stereochemistry whereapplicable.(a)…
A: (a) 1-methyl cycloheptene + BH3, THF, then H2O2, OH-(b) trans-4,4-dimethylpent-2-ene + BH3, THF,…
Q: he structure of the product formed in the reaction of an epoxide with ethoxide ion. If more than one…
A: We have to write the products.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- When Br2 is added to buta-1,3-diene at -15 °C, the product mixture contains 60% ofproduct A and 40% of product B. When the same reaction takes place at 60 °C, theproduct ratio is 10% A and 90% B.(a) Propose structures for products A and B. (Hint: In many cases, an allylic carbocationis more stable than a bromonium ion.)A conjugated diene with an even number of double bonds undergoes conrotatory ring closure underthermal conditions.Explain, with reference to the mechanism, why addition of one equivalent of HCl to diene A forms only two products of electrophilic addition, even though four constitutional isomers are possible.
- Highlight the allylic hydrogen(s) in this structure.In the dehydrohalogenation of bromocyclodecane, the major product is cis -cyclodecene rather than trans-cyclodecene. Offer an explanation.reaction of either 3-bromo-1-butene or (Z) -1-bromo-2-butene with water under SN1 condition yields the same product explain why
- The diene below when heated,will automatically cyclize into the cyclobutene shown . Explain how this reaction is possible accounting for the stereochemical outcomes(a) A compound known to be a substituted cyclohexanone derivative has lamda max of 235 nm. Could this compound be a conjugated dienone? explain (b) (i)For this compound, how many nm must be accounted for by substituents? (ii) What are the substituents and the points of substitution that may occur having accounted for the 20nm?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?
- Although dehydrohalogenation occurs with anti periplanar geometry,some eliminations have syn periplanar geometry. Examine the startingmaterial and product of attached elimination, and state whether theelimination occurs with syn or anti periplanar geometry.what product would you expect from photochemical cyclization of (2E,4z,6z)-2,4,6-octatriene with (MO) explinationRank the following dienophiles in order of increasing reactivity towards 1,3- butadiene Please explain

