Q: Phentermine was approved for sale in the United States in 2012 by the FDA. It is a psychostimulant d...
A: Intermediate 1 is known as diazonium salt...
Q: N2(g)+3H2(g)=2NH3(g) Following the reaction above, if 5.00 L of nitrogen reacts completely with hyd...
A:
Q: Write the equilibrium constant expression, Kc, for the following reaction: Please enter the compound...
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical eq...
Q: Be sure to answer all parts. At 430°C, the equilibrium constant (Kp) for the reaction 2NO(g) + O2(g)...
A: 2NO(g) + O2(g) ---> 2NO2(g) Kp = 1.5 × 105 Initial pressure of NO = 7.6 × 10-3 atm Initial pressu...
Q: The following chemical reaction takes place in aqueous solution: 2 AGNO,(aq)+K,CO3(aq) → Ag,CO3(s)+2...
A:
Q: Consider the equilibrium system described by the chemical reaction below. For this reaction, Kc = 2....
A:
Q: Consider the reaction 2 S(g) + 3 O2(g) → 2 SO3(g). Using the standard enthalpies of formation listed...
A: Here we have to determine the average bond energy of S=O from the given thermodynamic data using the...
Q: CHz Mg Br Ebz0 ,-40 °C 2) sto ag. Hz Soy
A: Here we are required to find the propose the mechanism for the addition reaction towards electrophi...
Q: your e Text. C6H5NH2 Write the formula for the conjugate acid of each of the following bases. Expres...
A: Write conjugate acid of C6H5NH2 ?
Q: For the following reaction between NH3 and CH3COOH, which one is the Bronsted-Lowry acid?
A:
Q: Calculate the normality of 0.321 g sodium carbonate when it mixes in a 250 mL solution.
A: Given, Mass of Sodium carbonate (Na2CO3) = 0.321 g Molat mass of Na2CO3 = 106 g/mol Mole = mass/MM ...
Q: HO. Ho So,H OEt
A: We have to predict the mechanism for given reaction.
Q: Part A Based on molecular structure, arrange the following oxyacids in order of increasing acid stre...
A:
Q: Consider the following reaction: 2503(g) 2s02(g) + 02(g) If 7.79x10-2 moles of So3(g), 0.491 moles o...
A: Equilibrium constant ( Kc ) is the ratio of concentration of products to reactants at equilibrium ra...
Q: Solutions that are electrolytes have ions in solution. These ions are surrounded by Tayers of water ...
A: In this question, we have to identify the ions from the compounds that will occur in the solution.
Q: Q: What is the difference between addition polymerization and condensation polymerization? In additi...
A: A polymer is a large molecule that is formed by the repetition of small units called a monomer, and ...
Q: 6. Which of the folloWing statements concerning ethanoic acid are correct? (1) It cannot be complete...
A: 6. Answer - The correct option is -(c) (2) and (3) only Explanation - According to the question - Gi...
Q: 12. Show how to convert cyclopentene into these compounds: ams bos Br (b) (a) Br (c) (d) OH Br
A: We have to predict the reagents for given conversions.
Q: Which of the following gas samples would have the largest volume, if all samples are at the same tem...
A:
Q: nat the time ol lloy(25%2n, 75%. cu) , submers ion in day of BYass avea was 10 cm², that 2.5 kg had ...
A:
Q: 2. A solution was prepared by dissolving 3.5 grams of ammonium chloride and 2 grams of ammonium hydr...
A: Given : Mass of ammonium chloride = 3.5 gm Mass of ammonium hydroxide = 2 gm Volume = 450 ml Si...
Q: Product: Nail Cutter 1. Why did we use metal in making nail cutterl? Why not ceramics or polymer? ...
A: The solution is shown below
Q: 2. Give the mechanisms and missing products for the reactions below. 1. 2. tertiary alcohol carbanio...
A: Detail mechanistic pathway is given below to find out the missing intermediate and final product
Q: For each system listed in the first column of the table below, decide (if possible) whether the chan...
A: Entropy means randomness. If the randomness or the motion of molecules increases in any case then th...
Q: What simple characterization techniques can you say that your product is dibenzalacetone?
A: Characterization techniques are the methods to identify, analyze the chemical substance and determin...
Q: a) NBS ROOR b) NBS ROOR c) NBS ROOR
A: NBS produces bromine radical and the order of stability of radicals is allylic>tertiary>second...
Q: Part A Identify the Brønsted-Lowry acid, the Brønsted-Lowry base, the conjugate acid, and the conjug...
A: We need to identify the Bronsted Lowry acid, base and the conjugate acid and base for the given reac...
Q: Aqueous solutions of barium chloride (BaCl) and sodium carbonate (Na2CO3) react to form solid barium...
A: Given: The volume of the 0.200 M barium chloride added = 100 mL The volume of the 0.150 M sodium car...
Q: A scientist removed a sample of 37.8 grams of a chemical from a container. The sample was 4 1/4 less...
A: Given the mass of the sample removed out of the total mass = 37.8 g Suppose the total mass of the c...
Q: a) 100 2-methyl-2-propanol 80- 40 20 0.0tt 10 20 30 40 50 60 70 80 m/z b) 100 n-propyl acetate 80 40...
A: Fragmentation of 2-methyl -2-propanol shows the elimination of small molecule OH , Methyl, H2O and ...
Q: A solution in prepared that has (NH,-1.30 M and a =0 105 M Part A How many grans of AgNO, can be dss...
A: Solubility product is the minimum ionic product of concentration of constituent ions of a salt requi...
Q: 4. Plexiglas is the trademark name of a strong, transparent polymer that is used to manufacture shat...
A: The monomer of the Plexiglas polymer which is used to manufacture shatter-resistant windows and shie...
Q: II. E,Z designation. A. Assign E or Z configuration to the following compounds. LOCH3 3. СНЗ 5. CHз ...
A: Definition :
Q: The electrolysis of molten AIClz for 4.25 hr with an electrical current of 25.0 A produces g of alum...
A: The amount of Aluminum produced can be calculated as follows
Q: A strong acid is a corrosive substance that can cause skin burns. If a strong acid is accidentally s...
A: Strong acid is substance which release hydrogen ions when dissolved in water. Strong base is substan...
Q: of the bonds C-C (single), C—C (double bond), and C—-C (triple bond), the C-C (single) bond is _____...
A: Bond length(pm) Bond energy(kJ/mol) C-C (single bond) 147 347 C=C (double bond) 134 614 C_=...
Q: Which side product/s do you expect in the adol condensation of acetone and benzaldehyde experiment? ...
A: Aldol condensation is condensation reaction between two molecules of same aldehyde or different alde...
Q: A cashew is burned in a soda can calorimeter, and heats up 0.245 kg of water by 3.65 °C. The calorim...
A: To calculate mass of fat contained in cashew , we would first Calculate heat given up by burning a c...
Q: When iron rusts, it undergoes a reaction with oxygen to form iron (II) oxide. 4 Fes + 30) 2Fe;Os (6)...
A:
Q: Consider the following reaction: 2HI(g) H2(g) + I2(g) If 4.13 moles of HI(g), 0.646 moles of H2, and...
A:
Q: What is the pH of 0.075 M methylammonium bromide, CH3NH3Br? (Kb of CH3NH2 = 4.4 × 10−4.)
A: The value of Kb for the given weak base methylammonium bromide is = 4.4x10-4 The concentration of th...
Q: allergic to the odor of ammonia. You smelled an ammonia solution with the pH 9.3 yesterday and it wa...
A:
Q: I Review | Constants | Periodic Rank the following compounds in order of decreasing acid strength us...
A: The order of acidity is given below
Q: cch the hyd of the helic
A: The amino acids can be classified based on the polarity of the side chains to form a scale called th...
Q: XA
A: The data given for Gibbs free energy and mole fraction of A at 1000°C (1273 K) for A-B solution is:
Q: What is the conjugate base of HCO3 ? Express your answer as a chemical formula. > View Available Hin...
A:
Q: hat the time ad submevs ion in day of Brass lloy(25%2n, 75%. cu) , avca was 10 cm², that 2. 5 kg had...
A:
Q: If 100.0 mL of potassium dichromate solution (10.0 g of K2Cr207 per liter) was mixed with 75.0 mL of...
A: First, we have to find the molarity of each of the K2Cr2O7(aq) or Cr2O72-(aq) and FeSO4.7H2O or Fe2+...
Q: Write out the net-ionic equation for the precipitation reaction that will happen with hydrogenphosph...
A:
Q: can you please do 4-6. show synthesis, synthons and disconnections OH но. 77 3. ÓH 4. ??? 777 5. 6.
A:
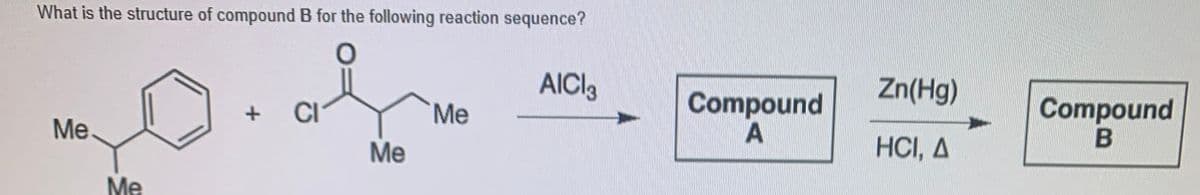
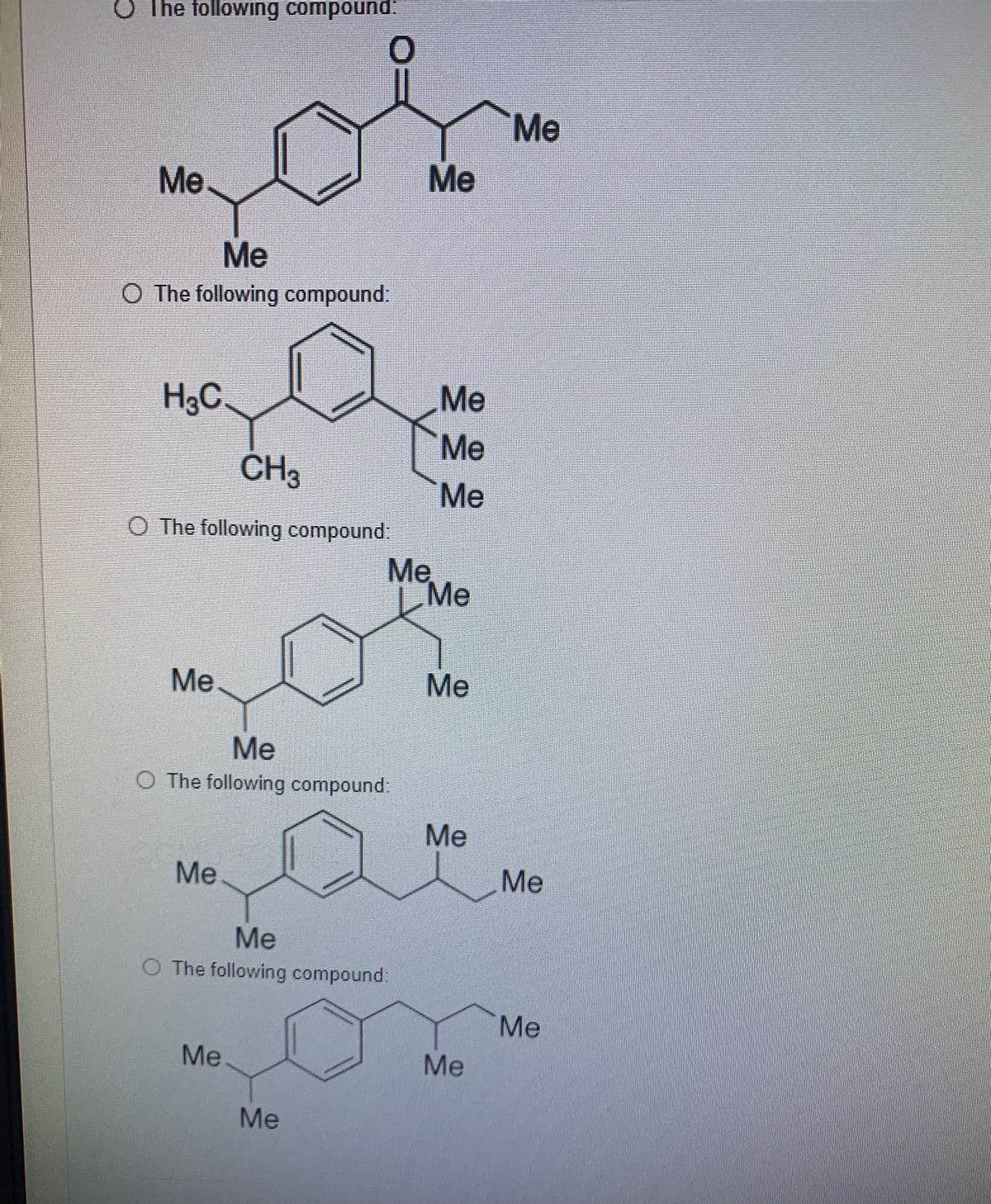
Step by step
Solved in 2 steps with 1 images

- Compound A is first reacted with methylamine in the presence of acid and then treated with NaBH3CN. Using the spectroscopic data given, what is the structure of the product after step 1?Name the structure. Heating compound A in the presence of H2SO4 results in which product?Identify the reagents represented by the letters a-e in the following scheme:
- The antipsychotic drug flupentixol is prepared by the following scheme:The (+) - aureol is a natural product that has shown selective activity against certain colon and lung cancer cell lines. It is postulated that a key step in the biosynthesis of this substance is the conversion of carbocation A into B. Propose a possible mechanism for this transformation (MUST show the corresponding arrows).Give the major organic product of the following reaction sequences (d & e).
- Propose a detailed mechanism for the transformations presented. represent the Intermediate structure A.Give the structure of the possible clasein condensation product from the following reaction. Tell which ,if any you would expect to predominate in each case. (a)CH3CO2Et+CH3CH2CO2Et (b)C6H6CO2Et + C6H5CH2CO2Et (c)EtOCO2Et +Cyclohexanones (d)C6H5CHo +CH3CO2EtGive the major organic product of the following reaction sequences (a-e).
- Explain the following observation. Ethyl 3-phenylpropanoate (C6H5CH2CH2CO2CH2CH3) reacts with electrophiles to afford ortho- and para-disubstituted arenes, but ethyl 3-phenylprop-2-enoate (C6H5CH= CHCO2CH2CH3) reacts with electrophiles to afford meta-disubstituted arenes.Identify compounds A – E of the reaction sequence shown in Scheme III, making sure to include stereochemistry as appropriate.a) Identify compound A in Scheme III. b) Identify compound B in Scheme III. c) Identify compound C in Scheme III. d) Identify compound D in Scheme III. e) Identify compound E in Scheme III.Identify compounds G and H in the following reaction scheme. H represents the structure of stemoamide, the chapter-opening molecule.

