ALEKS ← → C x A ALEKS - Tanvi Damle - Learn X 505 Missing Women.pdf X b Answered: A mixture of gaseou x + www-awu.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-lJjnzw4vpUz2IYCcR4NM7yr_IpMriNwxObE81fpfEarvknu9yVpdR16koytlCYH-ZAJTB3axbOp6jJjf_zb7s18YueB... ✩✩ ☐ ⠀ OTHERMOCHEMISTRY = 300 1/3 Tanvi V Understanding the definitions of heat and work 1 atm pressure A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gaseous products. The cylinder has a piston that moves in or out, as necessary, to keep a constant pressure on the mixture of 1 atm. The cylinder is also submerged in a large insulated water bath. (See sketch at right.) From previous experiments, this chemical reaction is known to absorb 298. kJ of energy. 18. gases The position of the piston is monitored, and it is determined from this data that the piston does 212. kJ of work on the system during the reaction. O exothermic Is the reaction exothermic or endothermic? 0 O endothermic O up S ? Does the temperature of the water bath go up on down? O down O neither Ⓒin Does the piston move in or out? O out Oneither Oin Does heat flow into or out of the gas mixture? O out O neither How much heat flows? Round your answer to 2 significant digits. X piston cylinder water bath olo
ALEKS ← → C x A ALEKS - Tanvi Damle - Learn X 505 Missing Women.pdf X b Answered: A mixture of gaseou x + www-awu.aleks.com/alekscgi/x/lsl.exe/10_u-IgNslkr7j8P3jH-lJjnzw4vpUz2IYCcR4NM7yr_IpMriNwxObE81fpfEarvknu9yVpdR16koytlCYH-ZAJTB3axbOp6jJjf_zb7s18YueB... ✩✩ ☐ ⠀ OTHERMOCHEMISTRY = 300 1/3 Tanvi V Understanding the definitions of heat and work 1 atm pressure A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gaseous products. The cylinder has a piston that moves in or out, as necessary, to keep a constant pressure on the mixture of 1 atm. The cylinder is also submerged in a large insulated water bath. (See sketch at right.) From previous experiments, this chemical reaction is known to absorb 298. kJ of energy. 18. gases The position of the piston is monitored, and it is determined from this data that the piston does 212. kJ of work on the system during the reaction. O exothermic Is the reaction exothermic or endothermic? 0 O endothermic O up S ? Does the temperature of the water bath go up on down? O down O neither Ⓒin Does the piston move in or out? O out Oneither Oin Does heat flow into or out of the gas mixture? O out O neither How much heat flows? Round your answer to 2 significant digits. X piston cylinder water bath olo
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.91PAE
Related questions
Question
Transcribed Image Text:←
X A ALEKS - Tanvi Damle - Learn X
505 Missing Women.pdf xb Answered: A mixture of gaseou x | +
с
www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-lgNslkr7j8P3jH-lJjnzw4vpUz2lYCcR4NM7yr_lpMriNwxObE81fpfEarvknu9yVpdR16koytlCYH-ZAJTB3axbOp6jJjf_zb7sl8YueB...
THERMOCHEMISTRY
=
1/3
Tanvi V
Understanding the definitions of heat and work
1 atm pressure
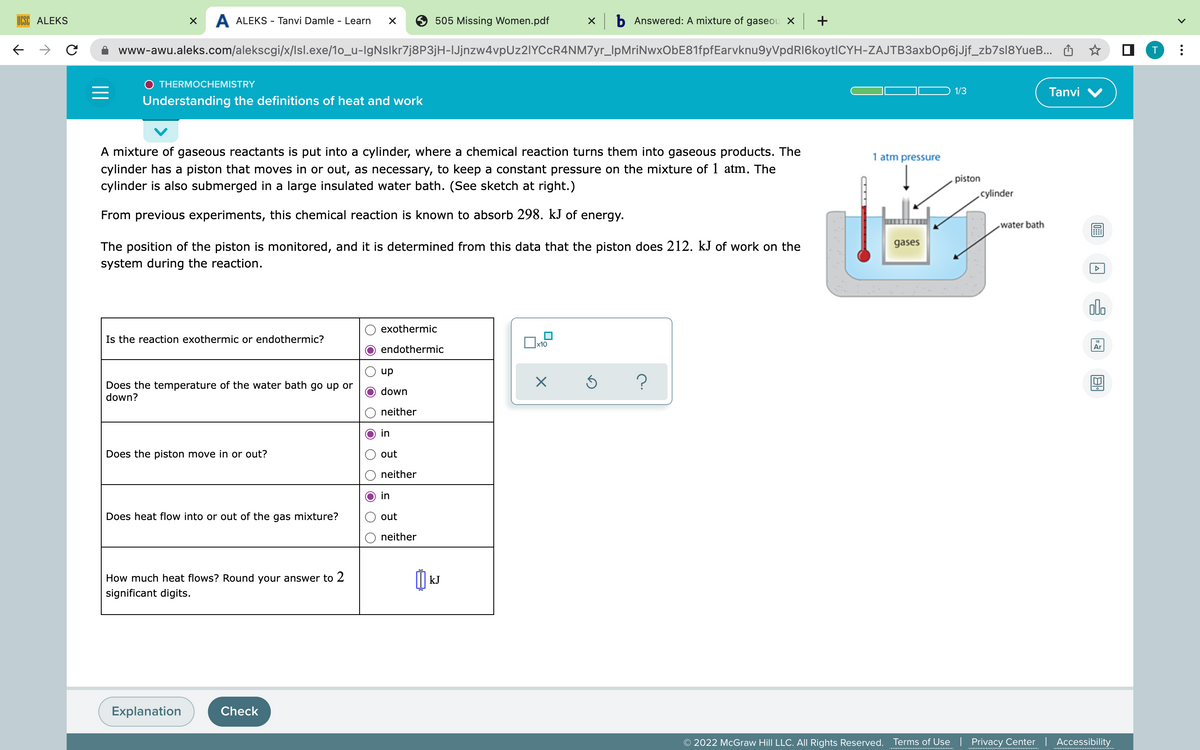
A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into gaseous products. The
cylinder has a piston that moves in or out, as necessary, to keep a constant pressure on the mixture of 1 atm. The
cylinder is also submerged in a large insulated water bath. (See sketch at right.)
From previous experiments, this chemical reaction is known to absorb 298. kJ of energy.
gases
The position of the piston is monitored, and it is determined from this data that the piston does 212. kJ of work on the
system during the reaction.
Is the reaction exothermic or endothermic?
exothermic
endothermic
x10
up
down
X
Does the temperature of the water bath go up or
down?
Ś ?
neither
in
Does the piston move in or out?
out
neither
in
Does heat flow into or out of the gas mixture?
out
neither
How much heat flows? Round your answer to 2
significant digits.
Explanation
Check
ALEKS
เม
kJ
piston
cylinder
water bath
0
A
olo
Ar
AL
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning