An aluminum bar of mass 2.00 kg at 330 °C is thrown into a lake. The temperature of the water in the lake is 12.0 °C; the specific heat capacity of aluminum is 900 J/(kg · K). Part D Since the aluminum bar is not an isolated system, the second law of thermodynamics cannot be applied to the bar alone. Rather, it should be applied to the bar in combination with its surroundings (the lake). What is the change in total entropy AStotal? Express your answer numerically in joules per Kelvin > View Available Hint(s) Π ΑΣφ ? AStotal J/K Submit
An aluminum bar of mass 2.00 kg at 330 °C is thrown into a lake. The temperature of the water in the lake is 12.0 °C; the specific heat capacity of aluminum is 900 J/(kg · K). Part D Since the aluminum bar is not an isolated system, the second law of thermodynamics cannot be applied to the bar alone. Rather, it should be applied to the bar in combination with its surroundings (the lake). What is the change in total entropy AStotal? Express your answer numerically in joules per Kelvin > View Available Hint(s) Π ΑΣφ ? AStotal J/K Submit
Physics for Scientists and Engineers: Foundations and Connections
1st Edition
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Katz, Debora M.
Chapter21: Heat And The First Law Of Thermodynamics
Section: Chapter Questions
Problem 31PQ: Consider the latent heat of fusion and the latent heat of vaporization for H2O, 3.33 105 J/kg and...
Related questions
Question
last 12 minutes. Please be fast
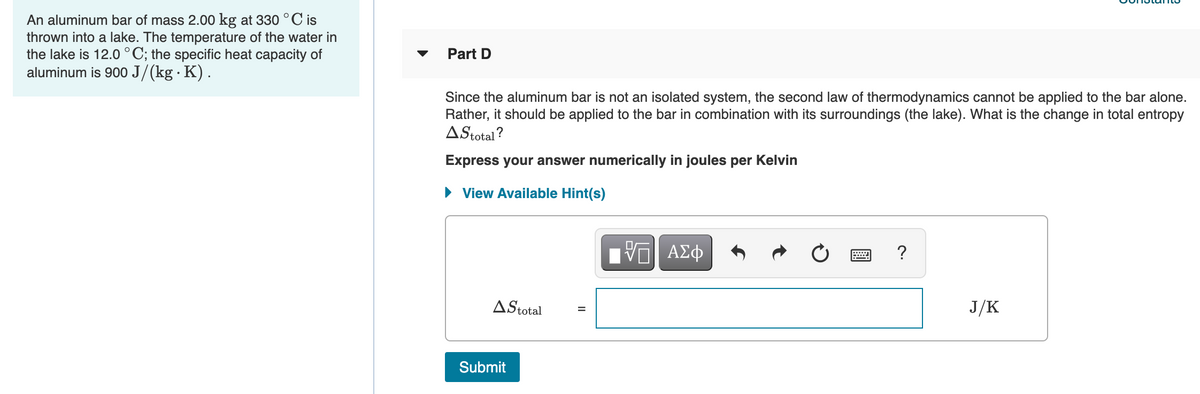
Transcribed Image Text:An aluminum bar of mass 2.00 kg at 330°C is
thrown into a lake. The temperature of the water in
the lake is 12.0 °C; the specific heat capacity of
aluminum is 900 J/(kg · K).
Part D
Since the aluminum bar is not an isolated system, the second law of thermodynamics cannot be applied to the bar alone.
Rather, it should be applied to the bar in combination with its surroundings (the lake). What is the change in total entropy
AStotal?
Express your answer numerically in joules per Kelvin
View Available Hint(s)
?
AStotal
J/K
Submit
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning