An amino acid structure contains a -COOH and -NH2 group in the same molecule. The -COOH group donates a proton to the -NH, group. Because of this, the amino acid structure contains a positive charge on one atom and a negative charge on the other. What is the term used to refer to this character of amino acids? O Amphiphatic O Isoelectric point O Zwitterions Amphoteric
An amino acid structure contains a -COOH and -NH2 group in the same molecule. The -COOH group donates a proton to the -NH, group. Because of this, the amino acid structure contains a positive charge on one atom and a negative charge on the other. What is the term used to refer to this character of amino acids? O Amphiphatic O Isoelectric point O Zwitterions Amphoteric
Biology: The Unity and Diversity of Life (MindTap Course List)
14th Edition
ISBN:9781305073951
Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Chapter3: Molecules Of Life
Section: Chapter Questions
Problem 3SQ: _______ groups are the acid part of amino acids and fatty acids. a. Hydroxyl (OH) b. Carboxyl (COOH)...
Related questions
Question
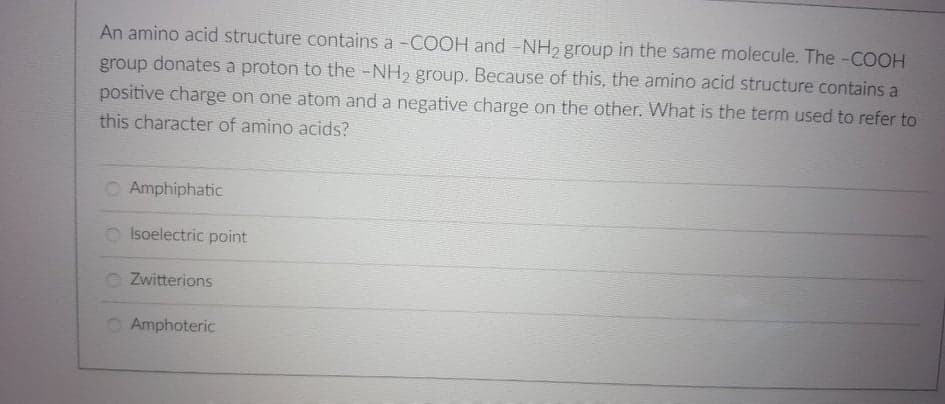
Transcribed Image Text:An amino acid structure contains a -COOH and -NH2 group in the same molecule. The -COOH
group donates a proton to the -NH2 group. Because of this, the amino acid structure contains a
positive charge on one atom and a negative charge on the other. What is the term used to refer to
this character of amino acids?
O Amphiphatic
O Isoelectric point
Zwitterions
O Amphoteric
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning

Biology: The Unity and Diversity of Life (MindTap…
Biology
ISBN:
9781305073951
Author:
Cecie Starr, Ralph Taggart, Christine Evers, Lisa Starr
Publisher:
Cengage Learning