An electro chemi cal cell is set up electrode with co pper CusO4 (ag) and 1.0 mol IL Pb (NO,)z caq) - in Contact with 1.0 mol L a lead Ucctrode in contact with the utandard reduction potentials ar Pbt t 2c- → ヤb E -0 13 V 2+ E°: +0-34V in At 25°C cell potential For thir galvanic cel the standcrd Vo Hs-
An electro chemi cal cell is set up electrode with co pper CusO4 (ag) and 1.0 mol IL Pb (NO,)z caq) - in Contact with 1.0 mol L a lead Ucctrode in contact with the utandard reduction potentials ar Pbt t 2c- → ヤb E -0 13 V 2+ E°: +0-34V in At 25°C cell potential For thir galvanic cel the standcrd Vo Hs-
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.6QAP
Related questions
Question
100%
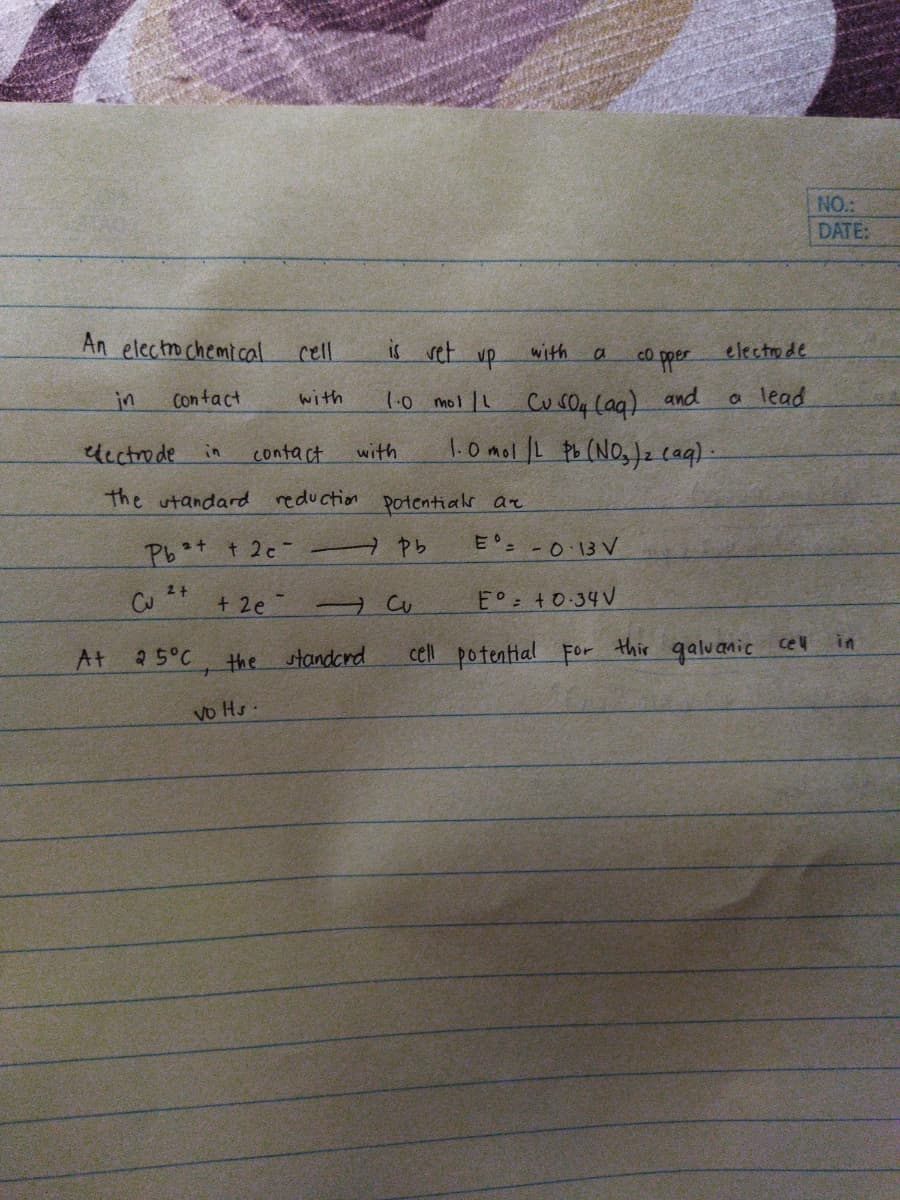
Transcribed Image Text:NO.:
DATE:
An electro chemical
is vet up
electrode
cell
with
co pper
Cu s04 lag) and
1.0mol L tb (NOs)z cag).
in
Contact
with
1.0 mol L
a lead
dcctrode
with
in
contact
the utandard reduction potentials ar
Pbt t 2c-
E° -0 13V
2+
E° +0-34V
thir galuanic
in
cel
At
25°C
the standcrd
cell potential For
vo Hs
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you



Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning



Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning