Q: Question 51 of 67 Identify the relationship between the following two compounds. gho A) identical B)…
A:
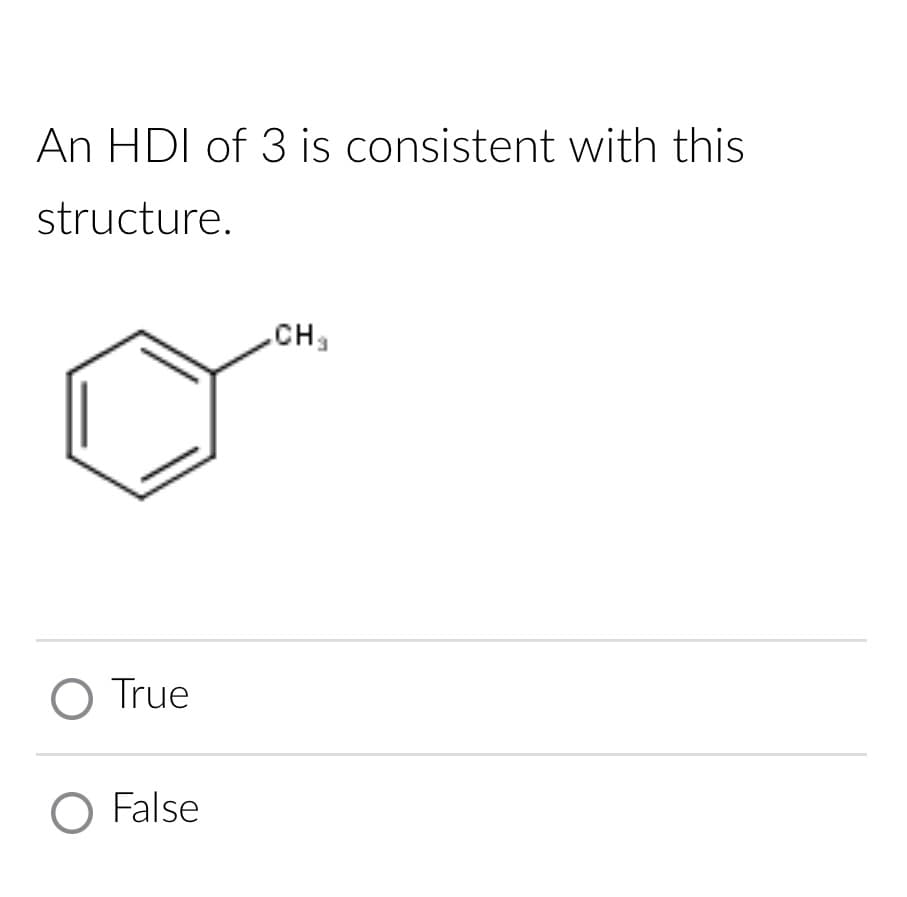
Q: d
A:
Q: What happens when oxalic acid reacts with potassium permanganate
A: Redox reaction takes place when oxalic acid reacts with potassium permanganate.
Q: For the following combustion reaction CH₃COCH₃(l) + 4O₂(g) → 3CO₂(g) + 3H₂O(g) ∆H = -1.79 × 10³ kJ…
A: This is exothermic reaction. one mole of acetone combustion leads to produce heat 1.79 × 10³ kJ in…
Q: A sample of argon gas occupies a volume of 6.03 L at 58°C and 446 mm Hg. The volume of the gas…
A:
Q: Determine the volume (in mL) of a 13.8 M HNO3 stock solution must be used to prepare 325.0 mL of a…
A: From the stock solution we can prepare the desired solution by diluting the solution upto the given…
Q: Identify which of the following compounds is meso.
A: Given : structure of molecules Tip : find out the R-S configuration
Q: CN. Rank the following compounds in order of reactivity toward an SN2 reaction. (1=best) CH3Br > 1…
A:
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A: When primary alkyl halide is treated with sodium acetate then substitution reaction occurs by SN²…
Q: Which of the following atom has a subshell which is half filled? O Ba O Fe O Au O Ar O Zn
A: The electron configuration of an element describes how electrons are distributed in its atomic…
Q: pH is a logarithmic scale used to indicate the hydrogen ion concentration, [H], of a solution: pH =…
A:
Q: would the molecular orbitals of cyclopenta-1,3-diene, the carbocation, carbanion, and the radical…
A:
Q: Measure out 50.0 mL 1.0 M CH3COOH and 50.0 mL of 1.0 M NaCH3COO and combine them into a beaker.…
A: A buffer solution is made up of 50.0 mL of 1.0 M CH3COOH and 50.0 mL of 1.0 M NaCH3COO, 2.0 mL of…
Q: Based on the type of electrolyte and concentration of the soulution, calculate the ion concentration…
A: The ion concentration in solution depends on the number of cations and anions it forms in solution…
Q: 3. Class 4 lasers can be used to remove tissue scars and to cut through strong metals. Why is this…
A: Lasers are divided into various groups from Class 1 to Class 4 based on the intensity of power…
Q: What quantity of heat (in kJ) is evolved when 25.0 g H2O(g) at 125.0 °C is converted to H2O(l) at…
A: we have to calculate the heat evolved when 25.0 g H2O(g) at 125.0 °C is converted to H2O(l) at 35.0…
Q: 1.00 mole of copper(II) sulfate, CuSO4, contains how many O atoms? O 2.41 x 1023 O atoms 2.41 x 1024…
A:
Q: go Consider the orbital shown. Indicate the number of radial and angular nodes in this orbital.…
A: The number of radial nodes can be calculated using the formula n–l–1. The number of angular nodes…
Q: Using this formula, you will explain some of the strange exceptions to the periodic trend of atomic…
A: 1) Atomic radius is the distance from an atom's nucleus to the outermost orbital of an electron.…
Q: At STP, what is the volume of 4.50 moles of nitrogen gas? 101 L O 34201 167 L 0607 L
A: Ant gas at STP condition ( standard temperature and pressure) has 22.4 L volume At STP condition…
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A: Given compound : 1. acetonitrile 2. sucrose 3. nitrous oxide
Q: Determine whether the molecules below are (R)- or (S)- isomers. C(CH3)3 H3COC Н НО. CH₂Br 0 Н""" H3C…
A: We have to first do the numbering according to the priority order. If it is clockwise and hydrogen…
Q: What quantity in moles of oxygen gas at 140.0 °C and 43.3 atm would occupy a vessel of 2.85 L?
A: temparature (T): 140.0 °C = 413 k pressure (P) = 43.3 atm volume (V)= 2.85 L gas constant (R) =…
Q: Consider the titration of 80.0 mL of 0.0200 M C6H5NH2 (a weak base; Kb = 4.30e-10) with 0.100 M…
A:
Q: 3er Server A3 Rab Rod Mayw lewat sepind dedor pod tyhle wei provided indicate the amount of rotation…
A:
Q: .4-Draw the structures of the following molecules: • a) trans-1-Bromo-3-methylcyclohexane .b)…
A: #4: (a): trans-1-bromo-3-methylcyclohexane: The longest carbon chain in the molecule is the…
Q: 7.79 We treat each chiral center separately. For each chiral center, we redraw one of the horizontal…
A: We give priority according to CIP rule. In this rule the group with highest atomic number is given…
Q: 0.100 mol of both H₂CO3 and KHCO3 are dissolved in 1.00 L of water. What reaction takes place when a…
A: In buffer solution, one is acidic part and another is basic part. So when an acid is added to…
Q: Choose the best reagents to complete the reaction shown below. mal H al A B D E 1. NaBH. 2. H₂O* Na…
A: Internal alkynes can be reduced into trans alkene when they react with sodium metal in liquid…
Q: Similarities I and M? I and C? M and C? Differences I and M? I and C? M and C?
A:
Q: You have a 0.50M stock of glucose, a 150mM stock of asparagine, and a 0.05M stock of NaH2PO4, and…
A:
Q: Problem IV: Reaction on Benzene Propose a synthesis for this compound starting from benzene: que…
A:
Q: VI. The rate of the reaction (CH3)3CCI+NaSCH2CH3 - ->> (CH3)2C=CH2 + CH3CH2SH + NaCl is first-order…
A: Given that a reaction is (CH3)3CCl + NaSCH2CH3 → (CH3)2C=CH2 + CH3CH2SH + NaCl. Also, given that the…
Q: Calcium magnesium acetate, which has the formula CaMg2(C2H3O2)6, is a popular de-icing agent. How…
A: We have calcium magnesium acetate, CaMg2(C2H3O2)6 We are to calculate the number of acetate ions in…
Q: The process of SDS-PAGE breaks peptide bonds. Select one: O True False
A: SDS-PAGE process breaks peptide bond??
Q: What is the pOH of a 0.0235 M HBr solution?
A:
Q: In a coffee cup calorimeter with a heat capacity of 21.5 J/ºC, 225 mL of 0.20 M KOH at 22.3 ºC…
A: Given : Volume of KOH = 225 ml Concentration of KOH = 0.20 M Volume of HCl = 225 ml Concentration…
Q: In the following reaction, what is the quantity of heat (in kJ) released when 4.93 moles of CH₄ are…
A:
Q: It is known that carbon dioxide (CO2) emission is responsible for at least 50% of greenhouse…
A: Carbon dioxide absorbs energy at a wavelength range of 2000 to 15000 nm's which are in infrared…
Q: If the molecule giving the ¹H NMR spectrum shown below has a total of 10 protons, how many protons…
A: In a proton NMR, the number of distinct hydrogens are represented by the number of peaks.
Q: 3 g of HA acid is dissolved in water to produce a 1 dm³ solution. If the change of acid…
A: Given, mass of acid, HA = 3 g Volume in dm3 = 1 dm3 Percent ionization = 2% Relative molecular…
Q: Question 4 Consider the following reaction and answer the questions that follow: MeO OMe…
A: Given that, an intramolecular claisen reaction scheme. We have to give the answers of the following…
Q: For each of the following processes, indicate whether the signs of S and H are expected to be…
A:
Q: For the following weak base, write a Bronsted Lowry acid base reaction with water showing the…
A: Bronsted Lowry base is a proton acceptor.
Q: Alkenes undergo an addition reaction with borane in tetrahydrofuran (THF). For the reaction below:…
A:
Q: Macmillan Learning A 28.01 g sample of a substance is initially at 23.9 °C. After absorbing 1229 J…
A: Mass = m = 28.01 g Heat = q = 1229 J Initial temperature = T1 = 23.9°C Final temperature = T2 =…
Q: Draw the curly arrow mechanism for the synthesis of furan using a diketone (Paal-Knorr synthesis of…
A: The Paal–Knorr Synthesis in organic chemistry is a reaction that generates either furans, pyrroles,…
Q: Which of the two samples has a higher mass in g? 10 mol He, or 0.2 mol Pb? O A. He B. Helium is a…
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to…
Q: H3C. Predict the 13C NMR chemical shift of the circled carbon in the structure below. VI H₂ CH3 A)…
A:
Q: At 25.0° C, a 10.00 L vessel is filled with 5.25 moles of Gas A and 4.70 moles of Gas B. What is the…
A: Given: Temperature of the system, T= 25.0°C = (25.0 + 273.15)K = 298.15 K Volume of the vessel, V…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- 1. Define sparging. What is the significance of this step in an HPLC analysis? 2. What are the two types of pre-column? What is the purpose of each type? 3. What are the two types of packing used in HPLC column? Describe each type. Please answer numbers 1-3. Need asapHow you can improve resolution in HPLC analysiswhich of the folowing(s) is/are true ? 1 split ratio in GC is a ratio of sample passed to column to amount of mobile phashe 2 column resolution decreases with increasing column lenght 3 decreasing the tube diameter ,increases the resolution
- A student performs a separation by TLC on a silica gel platee that results in three spots. A red spot is measured 23 mm away from the original sample spot, a blue spot is 42 mm away, and a yellow spot is 49 mm away. the distance from the original spot to where the solvent stopped is 55 mm. a) calculate the Rf value for each componenet in the mixture (show work and report th correct number of significant figures) b) the silica gel plate is polar. rank the three componenets in the mixture from lowest to highest polarity. Explain your answer.What is the balanced equation for the combustion of C8H7NO2SBrCl in aC,H,N,S elemental analyzer?Define the following terms used in HPLC: (d) sampling loops (e) bonded-phase packing (f) gel filtration
- In a normal phase chromatography set up, the component that yields the highest Rf value is likely to be _____. Group of answer choices Both polarity Cannot be determined Polar Non-polar2 d) In a typical HPLC column, 150 atm of pressure is required to push the mobile phase through the column. Why is high pressure needed in HPLC? e) Why are HPLC particles porous?It refers to the general term of adsorbent media used to visualize chromogenic compounds.A. ChromatogramB. Solvent Front C. Mobile PhaseD. Stationary Phase

