An ideal gas expands isothermally along AB and does 680 J of work. The process is illustrated in figure below (not drawn to scale). Answer below questions. PA a. How much heat does the gas exchange along AB? Net heat flow into gas is J. b. The gas then expands adiabatically along BC and does 480 J of work. When the gas returns to A along CA, it exhausts 105 J of heat to its surroundings. How much work is done on the gas along this path? Work done on the gas is J. c. What type of thermodynamic process does process CA represent? O neither isothermal nor adiabatic O isothermal process O adiabatic process O not enough information given for determination
An ideal gas expands isothermally along AB and does 680 J of work. The process is illustrated in figure below (not drawn to scale). Answer below questions. PA a. How much heat does the gas exchange along AB? Net heat flow into gas is J. b. The gas then expands adiabatically along BC and does 480 J of work. When the gas returns to A along CA, it exhausts 105 J of heat to its surroundings. How much work is done on the gas along this path? Work done on the gas is J. c. What type of thermodynamic process does process CA represent? O neither isothermal nor adiabatic O isothermal process O adiabatic process O not enough information given for determination
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 57P: An ideal gas expands isothermally along AB and does 700 J of work (see below). (a) How much heat...
Related questions
Question
#4
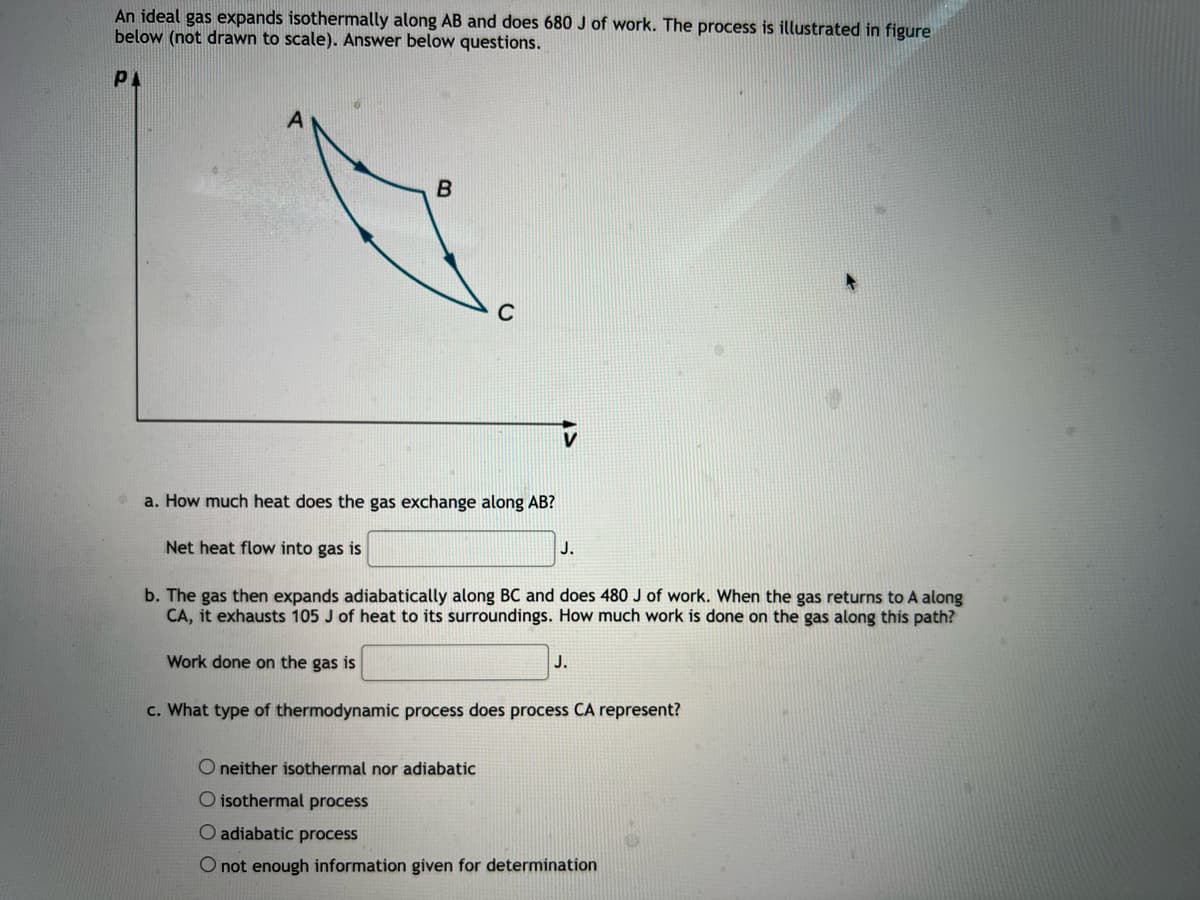
Transcribed Image Text:An ideal gas expands isothermally along AB and does 680 J of work. The process is illustrated in figure
below (not drawn to scale). Answer below questions.
PA
a. How much heat does the gas exchange along AB?
Net heat flow into gas is
J.
b. The gas then expands adiabatically along BC and does 480 J of work. When the gas returns to A along
CA, it exhausts 105 J of heat to its surroundings. How much work is done on the gas along this path?
Work done on the gas is
J.
c. What type of thermodynamic process does process CA represent?
O neither isothermal nor adiabatic
O isothermal process
O adiabatic process
O not enough information given for determination
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Physics for Scientists and Engineers with Modern …
Physics
ISBN:
9781337553292
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:
9781337553278
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers with Modern …
Physics
ISBN:
9781337553292
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers
Physics
ISBN:
9781337553278
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning