An impure sample contains 0.74 g of impurities and 4.55 g of benzoic acid. The sample is dissolved in 100 ml of water and heated. The solution is then cooled. How many grams of the impurity will crystallize when the solution has cooled? Include the unit and two decimal places in your answer. The solubilities of the impurity and benzoic acid in cold and hot water are given below. Compound Cold Water solubility Benzoic acid 0.42 g per 100 mL of water 5.79 g per 100 ml of water Impurity Hot Water Solubility 0.61 g per 100 mL of water 4.62 g per 100 mL of water
An impure sample contains 0.74 g of impurities and 4.55 g of benzoic acid. The sample is dissolved in 100 ml of water and heated. The solution is then cooled. How many grams of the impurity will crystallize when the solution has cooled? Include the unit and two decimal places in your answer. The solubilities of the impurity and benzoic acid in cold and hot water are given below. Compound Cold Water solubility Benzoic acid 0.42 g per 100 mL of water 5.79 g per 100 ml of water Impurity Hot Water Solubility 0.61 g per 100 mL of water 4.62 g per 100 mL of water
Chapter7: Solutions And Colloids
Section: Chapter Questions
Problem 7.5E
Related questions
Question
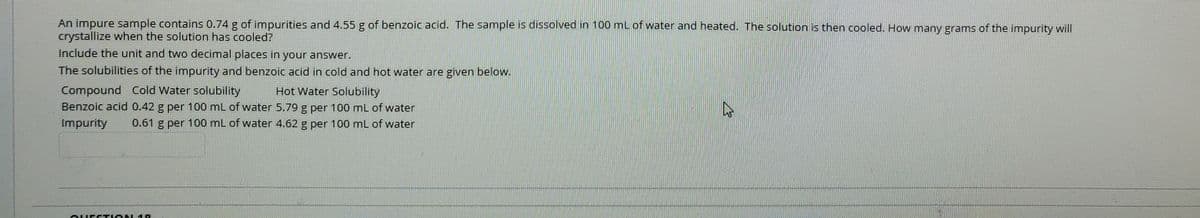
Transcribed Image Text:An impure sample contains 0.74 g of impurities and 4.55 g of benzoic acid. The sample is dissolved in 100 mL of water and heated. The solution is then cooled. How many grams of the impurity will
crystallize when the solution has cooled?
Include the unit and two decimal places in your answer.
The solubilities of the impurity and benzoic acid in cold and hot water are given below.
Compound Cold Water solubility
Benzoic acid 0.42 g per 100 mL of water 5.79 g per 100 mL of water
Hot Water Solubility
Impurity
0.61 g per 100 mL of water 4.62 g per 100 mL of water
OUESTIO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning