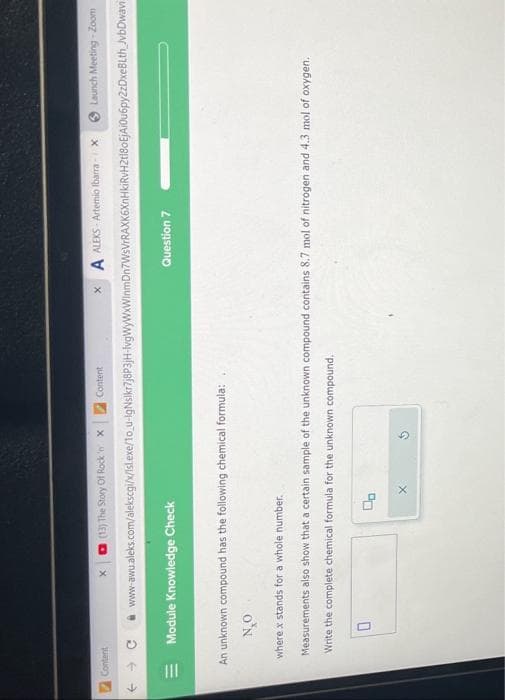
An unknown compound has the following chemical formula: N₂O where x stands for a whole number. Measurements Write the complete chemical formula for the unknown compound. 0 also show that a certain sample of the unknown compound contains 8.7 mol of nitrogen and 4.3 mol of oxygen. X
An unknown compound has the following chemical formula: N₂O where x stands for a whole number. Measurements Write the complete chemical formula for the unknown compound. 0 also show that a certain sample of the unknown compound contains 8.7 mol of nitrogen and 4.3 mol of oxygen. X
Chapter28: Atomic Spectroscopy
Section: Chapter Questions
Problem 28.4QAP
Related questions
Question
Transcribed Image Text:XA ALEKS-Artemio Ibarra-
C www-awu.aleks.com/alekscgi/x/isl.exe/1o_u-IgNslkr7j8P3jH-fvgWyWxWinmDn7WsVrRAXK6XnHkiRvH2tl8oEjAi0u6py2zDxeBLth_JvbDwavi
Content
X
(13) The Story Of Rock 'nx Content
Module Knowledge Check
An unknown compound has the following chemical formula:
NO
0
Question 7
Launch Meeting - Zoom
where x stands for a whole number.
Measurements also show that a certain sample of the unknown compound contains 8.7 mol of nitrogen and 4.3 mol of oxygen.
Write the complete chemical formula for the unknown compound.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

