ANALYSIS. Five unknown isomeric compounds were found to have the molecular formula C5H100. Upon Clemmensen reduction, compounds A, B, and C all yielded n-pentane. These compounds were further subjected to chemical tests to determine their identity. The results of the chemical tests are shown in the table: TEST RGT Cpd A Cpd B + + + Cpd C Cpd D Cpd E + Phenylhydrazine Tollen's rgt 12, КОН CrO3, H+ Based on the test results, draw the structures of compounds A to E. + + +
ANALYSIS. Five unknown isomeric compounds were found to have the molecular formula C5H100. Upon Clemmensen reduction, compounds A, B, and C all yielded n-pentane. These compounds were further subjected to chemical tests to determine their identity. The results of the chemical tests are shown in the table: TEST RGT Cpd A Cpd B + + + Cpd C Cpd D Cpd E + Phenylhydrazine Tollen's rgt 12, КОН CrO3, H+ Based on the test results, draw the structures of compounds A to E. + + +
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter20: Dienes, Conjugated Systems, And Pericyclic Reactions
Section: Chapter Questions
Problem 20.38P
Related questions
Question
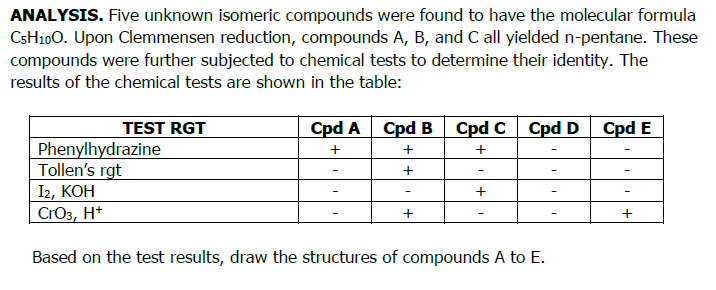
Transcribed Image Text:ANALYSIS. Five unknown isomeric compounds were found to have the molecular formula
C5H100. Upon Clemmensen reduction, compounds A, B, and C all yielded n-pentane. These
compounds were further subjected to chemical tests to determine their identity. The
results of the chemical tests are shown in the table:
TEST RGT
Cpd A Cpd B Cpd C Cpd D
+
+
Phenylhydrazine
Tollen's rgt
I2, KOH
CrO3, H+
Based on the test results, draw the structures of compounds A to E.
+
+
+
+
Cpd E
+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning