Analyst Date 1% 102- 95- 90- 85- 80 75 70- 65- 60- 55- 50- 45 40 35- 30 28- 4000 Sample Name Administrator 06 3500 Administrator Thursday, March 2, 2023 1:44 PM 2862.16cm-1 2935.42cm-1 3000 2500 1703.53cm-1 cm-1 2000 Description Sample 006 Bv Administrator Date Thursday, March 02 2023 1449.28cm-1 1500 PerkinElmer Spectrum IR Version 10.7.2 Thursday, March 2, 2023 1:44 PM pmmm 749.83cm-1 1310.91cm-1 908.06cm-1 1221.50cm-1 1000 489.08cm 1118.37cm-1 500450 Quality Checks The Quality Checks do not report any warnings for the sample.
Analyst Date 1% 102- 95- 90- 85- 80 75 70- 65- 60- 55- 50- 45 40 35- 30 28- 4000 Sample Name Administrator 06 3500 Administrator Thursday, March 2, 2023 1:44 PM 2862.16cm-1 2935.42cm-1 3000 2500 1703.53cm-1 cm-1 2000 Description Sample 006 Bv Administrator Date Thursday, March 02 2023 1449.28cm-1 1500 PerkinElmer Spectrum IR Version 10.7.2 Thursday, March 2, 2023 1:44 PM pmmm 749.83cm-1 1310.91cm-1 908.06cm-1 1221.50cm-1 1000 489.08cm 1118.37cm-1 500450 Quality Checks The Quality Checks do not report any warnings for the sample.
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
Please help me to interpret this spectrum.
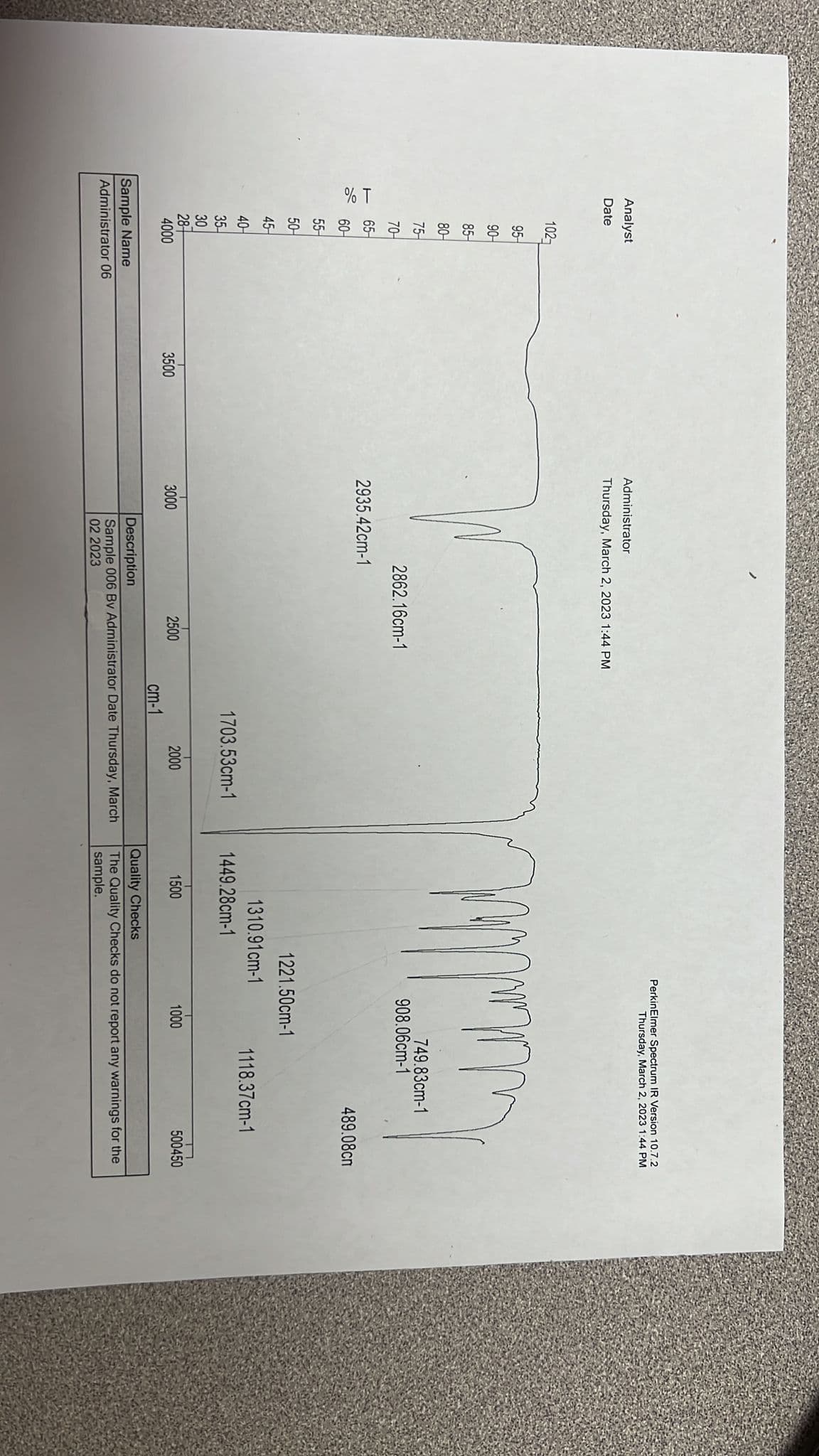
Transcribed Image Text:Analyst
Date
% T
§-s † ‡ † ¢ ¢ ¢ ¦† †¶¶¶ &
4000
Sample Name
Administrator 06
3500
Administrator
Thursday, March 2, 2023 1:44 PM
2862.16cm-1
2935.42cm-1
3000
2500
1703.53cm-1
cm-1
2000
Description
Sample 006 Bv Administrator Date Thursday, March
02 2023
1449.28cm-1
PerkinElmer Spectrum IR Version 10.7.2
Thursday, March 2, 2023 1:44 PM
1500
mmm
1310.91cm-1
1221.50cm-1
908.06cm-1
749.83cm-1
1000
489.08cm
1118.37cm-1
500450
Quality Checks
The Quality Checks do not report any warnings for the
sample.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning