Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.32QAP
Related questions
Question
! ( give ans with explanation ) if u hv answerd earlier plz do not answer because earlier answer was wrong. give only correct answer with explanation
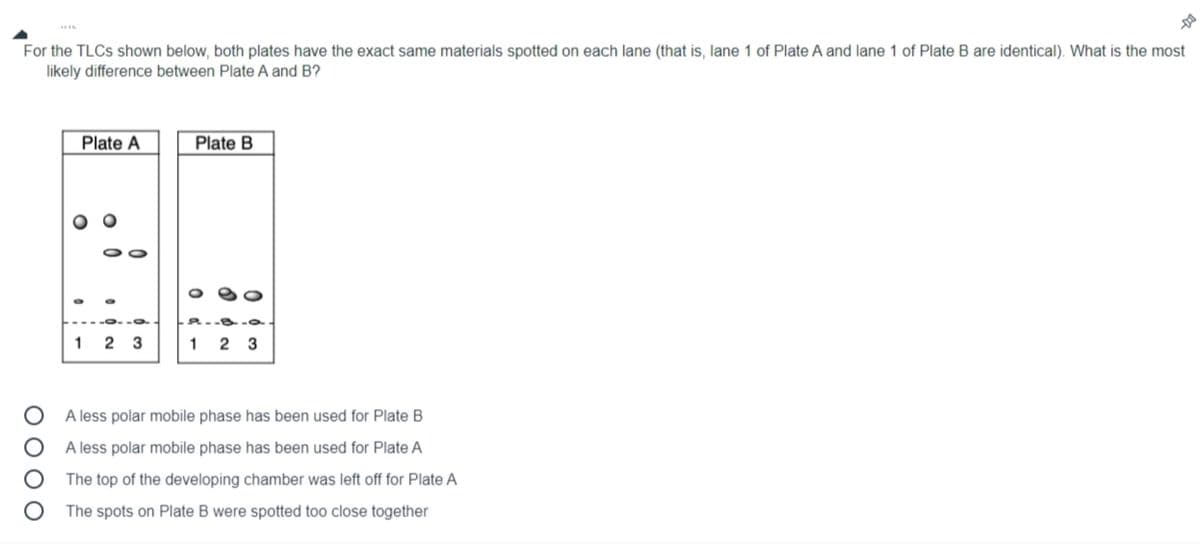
Transcribed Image Text:For the TLCS shown below, both plates have the exact same materials spotted on each lane (that is, lane 1 of Plate A and lane 1 of Plate B are identical). What is the most
likely difference between Plate A and B?
Plate A
Plate B
1 2 3
1 2 3
A less polar mobile phase has been used for Plate B
A less polar mobile phase has been used for Plate A
The top of the developing chamber was left off for Plate A
The spots on Plate B were spotted too close together
O O O O
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
