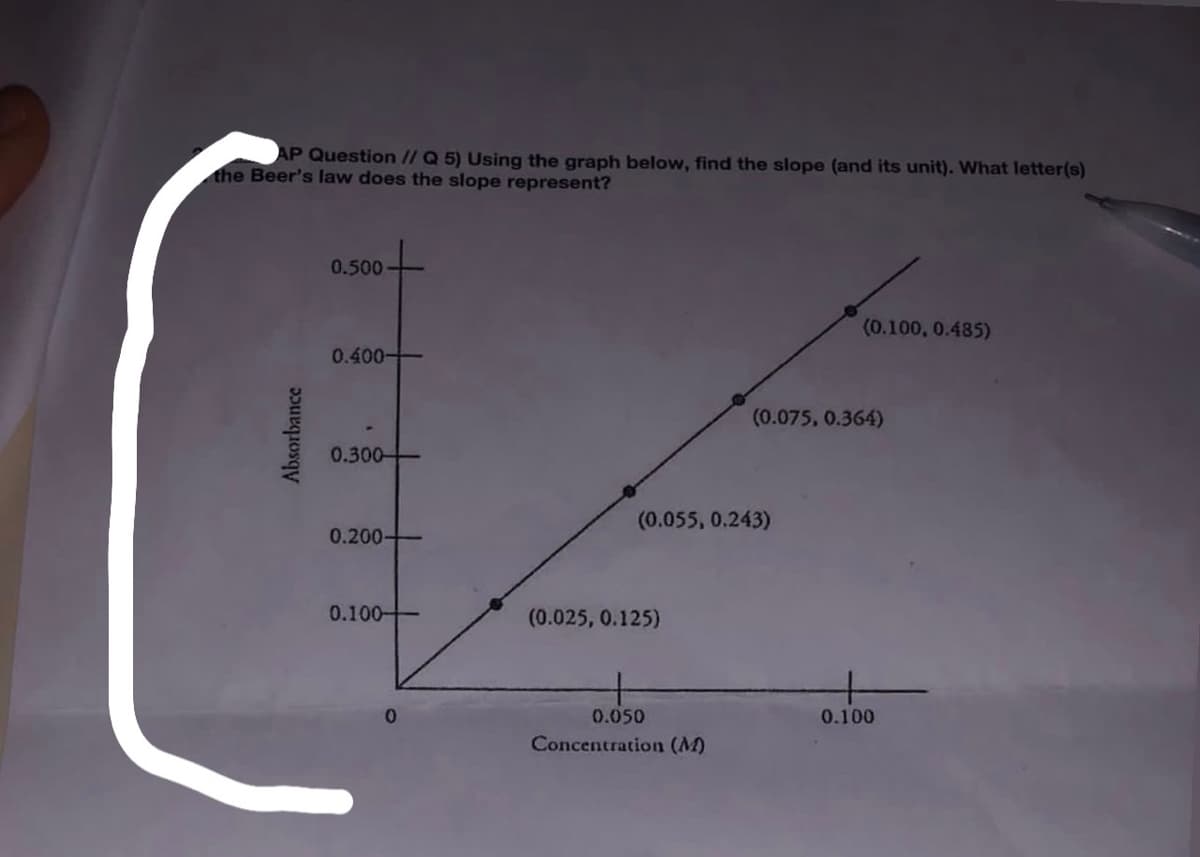
AP Question // Q 5) Using the graph below, find the slope (and its unit). What letter(s) the Beer's law does the slope represent? 0.500 (0.100, 0.485) 0.400 - (0.075, 0.364) 0.300- (0.055, 0.243) 0.200- 0.100- (0.025, 0.125) 0.050 0.100 Concentration (M) Absorbance
Q: What is the reaction mechanism?
A: Here we have to write mechanism of the following synthesis reaction.
Q: Carbonyl chloride (COCI,), also called phosgene, was used in World War I as a poisonous gas. The equ...
A: Given :- CO(g) + Cl2(g) <--> COCl2(g) [CO] = 0.0290 M [Cl2] = 0.0400 M [COCl2] = 0.255 M T...
Q: Tell whether the given reaction will occur via SN1, SN2, E1, or E2
A: In the given reaction the given alkyl fluoride gives an acetolysis reaction with acetic acid. The al...
Q: NH Oit 1d 25 1d 1s 25 4t 10 8. 7 6 5 1. 2. 3. 1.
A:
Q: Calculate the concentrations of all species present in a 0.18 M solution of ethylammonium chloride (...
A: Given, the concentration of C2H5NH3Cl = 0.18 M Ka for C2H5NH3+ = 1.8×10-11 We have to calculate the ...
Q: A 10.0 g sample of sodium metal reacts with 2.50 L of nitrogen gas at 0.976 atm and 28∘C to produce ...
A: Balanced chemical equation can be defined as the equation in which equal number of atoms of each ele...
Q: Consider the following system at equilibrium where AH° = 18.8 k), and Kc = 9.52x10-2, at 350 K: CH4 ...
A: Le Chatelier's principle: This principle is used to predict the effect of a change in conditions on ...
Q: The solubility of lead(II) iodide is 1.39 x 10-3 mol/L at a certain temperature. What is the solubil...
A:
Q: question 12 Calculate the initial molarity of a generic amine, R3N (Kb = 7.23x10-6), whose pOH = 2....
A: Here we have to calculate the initial molarity of a generic amine, R3N where we have given that the ...
Q: 5. Give the reagent(s), conditions etc. needed to selectively protect the substrates below to the de...
A:
Q: 7. The volume of a gas-filled balloon is 50.0 L at 20.0∘C and 742 torr. What volume will it occupy a...
A:
Q: The half-life of a second-order reaction does not depend on the initial concentration of reactant. T...
A:
Q: Provide structures for aromatic hydrocarbons that meet the following criteria: a. compound C9H12 tha...
A: The given reactions are electrophilic substitution reactions namely halogenation reaction and these ...
Q: Be sure to answer all parts. Consider the following equilibrium at 447.9 K: NH,HS(s) S NH3(g) + H,S(...
A: In the given questions, We first need to calculate Kp since partial pressure of gases is given.Then...
Q: Calculate the solubility (in g/L) of CaSO,(s) in 0.450 M Na, SO, (aq) at 25°C. The Ksp of CaSO, is 4...
A:
Q: Consider the equilibrium system described by the chemical reaction below. If the partial pressures a...
A:
Q: by the reaction of hydrochloric acid with manganese(IV) oxide. HCl(aq) + MnO,(s) - MnCl, (aq) + 2 H,...
A: 4HCl (aq) + MnO2(s) ----> MnCl2(aq) + 2H2O (l) + Cl2(g) Mass of HCl = 44.1 g Mass of MnO2 = 43.7 ...
Q: OH OH CH
A:
Q: Consider the reaction: Mg(OH)2 (s) = Mg²+ (aq) + 2 OH (aq) At equilibrium, a 1.0 L reaction vessel c...
A:
Q: For the following molecules, list them from low boiling point to high boiling point, and list the st...
A: SiH4 PH3 CH3OH
Q: The equilibrium constant, K, for the following reaction is 1.20x10-2 at 500 K. PCI5(g) PCI3(g) + Cl2...
A: Kc is the ratio of product of concentration of products raised to their stoichiometric coefficient ...
Q: Sulfuryl chloride, SO2C12(9), decomposes at high temperature to form SO2(9) and Cl2(g). The rate con...
A: Given :- SO2Cl2(g) → SO2(g) + Cl2(g) Rate constant (K) = 4.68 × 10-5 s-1 To determine :- Order o...
Q: Identify the battery that is in most automobiles. NiCad battery lithium-ion battery fuel cell lead-a...
A: Automobiles are of various kinds and hence the kind of batteries used in automobiles will also be of...
Q: Calculate the standard entropy change for the reaction at 25 °C. C;H,(g) + 50,(g) • 3 CO,(g) + 4 H,O...
A:
Q: Match the type of electromagnetic radiation with its frequency. gamma rays v [ Choose ] 10E15 Hz 10E...
A:
Q: A wave of blue light has a frequency of 7.65x1014 s-1. What is the energy of each light wave? Use h...
A: Given, Frequency of light wave (v)= 7.65x1014 s-1 Planck constant (h) = 6.626x10-34 J•s Energy of li...
Q: What the time l submevs ion in day ot Brass alloy (25%2n, 45%. cu) , avea was 10 cm2, that 2.5 Kg ノ ...
A: Given, Brass alloy having 25%zinc and 75%copper Area-10cm2 Weight of NACL displaced by brass-2.5kg ...
Q: Illustrate the electron spin quantum number of Boron
A: Quantum mumbers are used to describe position of electrons in orbitals of an atom. There are four di...
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of t...
A: Compound can be defined as a chemical substance composed of molecules that consist of atoms of two ...
Q: What's the partial pressure of carbon dioxide in a container that holds 5 moles of carbon dioxide, 3...
A: Calculate partial pressure of CO2 in the mixture ?
Q: Magnesium phosphate is an ionic compound which is widely used in medication to support the relaxatio...
A: The formula of ionic compounds is written by following electrical neutrality principle, which states...
Q: What is the pH of 0.075 M methylammonium bromide, CH3NH3Br? (Kb of CH3NH2 = 4.4 × 10−4.)
A: The value of Kb for the given weak base methylammonium bromide is = 4.4x10-4 The concentration of th...
Q: Me Me D Me Me
A: Here we are required to find the reagent D which is used to form metal This is done by reterosynthes...
Q: are spectator ions?
A:
Q: Calculate the concentration of H3O* in a solution that contains 5.5 x 10-5 M OH at 25°C. Identify th...
A: For an aqueous solution, we know that the production of the concentration of the hydrogen ions and t...
Q: Part A A soluton is prepared that has NI-130 M and (CI"-0.100 M. How many grams of AgNO, can be diss...
A: Solubility product is the minimum ionic product of concentration of constituent ions of a salt requi...
Q: How many kWh of energy does a 600-W toaster use in the morning if it is in operation for a total of ...
A:
Q: gaseous mixture contains 4.56 g propane, C3H8, and 1.22 g chloroform, CHCL3. Assuming that both comp...
A: Mass of propane = 4.56 g Mass of chloroform = 1.22 g V = 100 ml = 0.1 L T = 28oC = 301 K Molar ma...
Q: For each system listed in the first column of the table below, decide (if possible) whether the chan...
A: Entropy means randomness. If the randomness or the motion of molecules increases in any case then th...
Q: ne gases dinitrogen monoxide, N 0, and propane, 38 nd pressure, have the same number of atoms. Answe...
A:
Q: Be sure to answer all parts. The equilibrium constant (K) for the reaction 2HCI(g) - H,(g) + Cl,(g) ...
A: What is the equilibrium constant of the required reaction---
Q: Calculate the AG value for the oxidation of succinate using NAD*. AG" 68 kl/mol Calculate the AGe va...
A: Free energy change gives the spontaneity of a reaction.
Q: Which of the following processes have a AS > 0? CH3OH(I) – CH3OH(s) O N2(8) + 3 H2(g) → 2 NH3(g) CHĄ...
A:
Q: 4. How much heat is released when 29.5 grams of Cl2 (g) reacts with excess hydrogen? H2 (g) + Cl2 (g...
A: Given, H2(g) + Cl2(g) ➝ 2HCl(g) ∆H° = -186 kJ Note: negative sign indicate the release of energy as...
Q: (c) Estimate the wavelength of electrons that have been accelerated from rest through a potential di...
A: An electron is a negatively charged particle.Wavelength can be defined as the distance from one cres...
Q: solution is prepared by dissolving 37.3 g of potassium chloride in distilled water. What is the volu...
A: A) Molarity (M) = 2.0M Mass = 37.3 g Molar mass of KCl = 74.6 g/mol Mole = mass/molar mass = 37.3...
Q: Question 10 Choose the correct product for the Diels-Alder reaction shown: A C A
A:
Q: How many H2 molecules are needed to completely hydrogenate the following triacylglycerol molecule? (...
A:
Q: A cylinder contains helium gas with a volume of 2.60L at 20°C and 768 mmHg. If the balloon ascends t...
A: Given:: V1=2.60 L P1=768 mmHg P2=614 mmHg V2= ?
Q: (c) Estimate the wavelength of electrons that have been accelerated from rest through a potential di...
A: In the given question we have to calculate the wavelength of electrons.
Step by step
Solved in 3 steps with 1 images

- pH = 11.7 Express your answer using one significant figure. [H3O+][H3O+] = MMDr. Colibrí studies hummingbirds. Each bird requires approximately 9.52 milliliters of nectar every day to stay alive. Synthetic nectar solution is made by mixing 20%(vol/vol) sucrose with 80%(vol/vol)water. Sucrose, common table sugar, has a density of 1.59 g/mL. How many grams of sugar does Dr. Colibri need to keep 7 hummingbirds alive for 63 days? (answer to 2 decimal places) Assumption: When mixing 1 part sugar with 4 parts water, the combined volume does not change. e.g. 200 mL of sugar combined with 800 mL water will produce 1000 mL of nectar. This is technically untrue, but it is close enough for our purposes.Question 11 .A sample of Fluorine occupies 65.5 mL at 23.0 oC. Calculate the final volume in mL when the temperature increased to 62.5oC. The final answer is... use the G.U.E.S.S. Method format. V2 = Unit = Blank 1: Blank 2: Please make sure that you convert all temperature readings to Kelvin (K = C + 273). Please make sure to double and triple check your sigfig when entering your answers. All answers should be entered in standard notation (not scientific notation). Please be sure to capitalized the correct letters in your units.
- Write the -log of 1.0, 0.1, 0.01, 0.001 and 0.0001 (it might help to make a neat table). Which range of values listed includes the value of the -log 0.005? a. between -5 and -6 b. Between 2 and 3 c. Between 1 and 2 d. Between 3 and 4 e. between -4 and -5Plot 1/λ (y-axis) vs 1/n2 ( x-axis) for hydrogen and attach your graph to the back of this handout. Excel graphs are not permitted. Your graph should have: a titie, proper axis scaling, axes labelled including units, data points shown clearly eg. dot in a circle, best straight line drawn, and the coordinates (x,y) of the points on the straight line chosen to calculate the slope. Never use data points to calculate the slope! Show slope calculation directly on the graph. 3. a) From your graph, calculate the experimental value of R, the Rydberg constant. (Note - this is the absolute value of your slope): b) Based on the theoretical value of R,1.0974×107 m−1, calculate your % error: information you need to solve this question: The picture is attachedHow many mL of 12% SDS would you need to make 23 mL of 2 % SDS? Report your answer to 1 decimal place (example: 3.4
- Does anyone know how to answer this question: Please helpWhat is the percentage change in x in going from x1 to x2 (%∆x)? a) 100(∆x1/x) b) 100(∆x2/x) c) 100(∆x/x1) d) 100(∆x/x2) e) none of the aboveStan Moneymaker needs 15 gallons of gasoline to top off his automobile’s gas tank. If he drives an extra eight miles (round trip) to a gas station on the outskirts of town, Stan can save $0.10 per gallon on the price of gasoline. Suppose gasoline costs $3.90 per gallon and Stan’s car gets 25 mpg for in-town driving. Should Stan make the trip to get less expensive gasoline? Each mile that Stan drives creates one pound of carbon dioxide. Each pound of CO2 has a cost impact of $0.02 on the environment. What other factors (cost and otherwise) should Stan consider in his decision making?from lowest to highest, 4 being the lowest
- 14 - For CH4 molecule containing avagadro’s number of atoms; I. It is 0,2 moles. II. Contains 4,8x1023 pieces of H atoms. III. It is 4,48 liters in UNC. Which of the statements are corrrect? A) II and III B) Only II C) I and II D) I, II and III E) Only IA previous Chem 100 student carried out the experiment you did by reacting 0.031 g of magnesium. The following data was collected in their lab notebook. The water temperature was 19.8°C. The barometer in the lab read 753 mm Hg and 31.97 mL of gas was collected The vapor pressure of water at this temperature is 17.32 mm Hg. Calculate the student’s experimental value of R. Report your answer to four places after the decimal.During Normal breathing while resting Abigails lungs contain 751.6mm Hg in 3.1L after inhaling and 762.3 mm Hg in 2.5L after exhaling. If her lungs conatin 0.114 Mol of air after inhaling how many moles if air remain after she exhales? Assume the temperature stays constant 37 degrees Type your answer to the three places past the decimal

