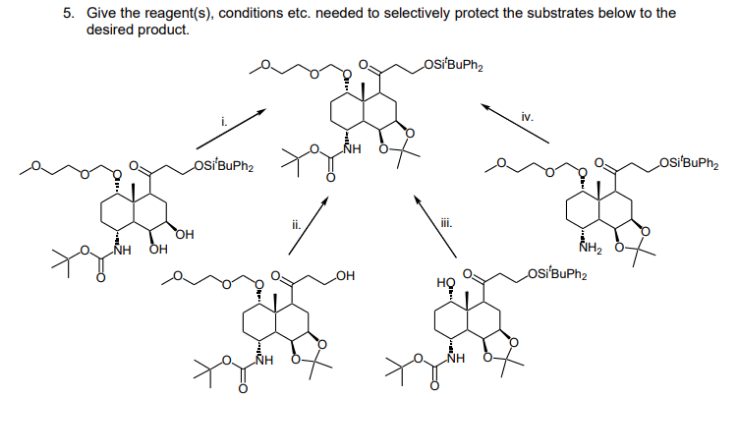
5. Give the reagent(s), conditions etc. needed to selectively protect the substrates below to the desired product. OSÍBUPH2 iv. osÍBuPh2 osiBuPh2 ii. i. OH NH osIBuPh2 NH
Q: Consider the following reaction where K. = 1.29x10 2 at 600 K. COCI,(g) cO(g) + Cl2(g) A reaction mi...
A:
Q: cch the hyd of the helic
A: The amino acids can be classified based on the polarity of the side chains to form a scale called th...
Q: Calculate the pH of a mixture that contains 0.21 M of HCOOH and 0.12 M of HCIO. The K, of HCOOH is 1...
A:
Q: When NH3(g) reacts with N20(g) according to the following reaction, 107.3 kcal of energy are evolved...
A:
Q: Part A Draw the condensed structural formula of the amine salt formed when lidocaine reacts with HCI...
A: GIVEN:- Draw the condensed structural formula of the amine salt formed when lidocaine reacts HCI.
Q: Calculate the standard free-energy change of the reaction catalysed by the enzyme phosphoglucomutase...
A: The solution is given below -
Q: Compare the two quantities based on the given condition: Titration of 25 mL 0.01 M NaOH with HBr I...
A: Given : Volume of NaOH = 25 ml Concentration of NaOH = 0.01 M Concentration of HBr in I case = 0....
Q: Gaseous SO2Cl2 reacts with liquid water to form hydrogen chloride gas and liquid sulfuric acid. Usin...
A: Given: Gaseous SO2Cl2 reacts with liquid water to form hydrogen chloride gas and liquid sulfuric aci...
Q: his Diagram or cycle The Biomass Pyrolysis-Cycle BIOMASS Gas recycle Heat for drying Dry GAS Grind P...
A:
Q: mma my mml my be able to mue Diameter of the FOV: mm differen wo types of 1500 micrometers (um). din...
A:
Q: Calculate the change in entropy associated with the Haber process to produce ammonia from nitrogen a...
A: Given, S°(NH3) = 192.5 J/mol-K S°(N2) = 191.5 J/mol-K S°(H2) = 130.6 J/mol-K □Srxn = [Total entropy...
Q: Calculate the pH of a solution that contains 3.9 x 104 M H3O* at 25°C. 4.59 3.41 10.59 9.41 0.59
A: Given, Concentration of H3O+ ion = [H3O+] = 3.9 × 10-4 M Temperature (T) = 25°C pH of the solution =...
Q: Consider the following reaction where K, = 10.5 at 350 K. 2CH2CI2(g) CH4(9) + CCI4(g) A reaction mix...
A:
Q: The solubility of lead(II) iodide is 1.39 x 10-3 mol/L at a certain temperature. What is the solubil...
A:
Q: Use the data below to calculate AG°rxn for the reaction: H, (g) +CO,(g) – H,0(g) +CO(g) Data: H, (g)...
A:
Q: Calculate the normality of NaOH solution formed by dissolving 0.2 gm NaOH to make 250 ml solution.
A: Normality of solution is defined as number of gram equivalents of solute dissolved per litre of solu...
Q: Mg, ether SOCI, Br H,O с. HO, d.
A:
Q: 1. The pH of a 0.1 M solution of a monobasic acid P is 1.0 while the pH of a 0.2 M solution of anoth...
A: Introduction : pH can be defined as a measure of the hydrogen ion concentration of a solution. Solu...
Q: Part A: The volume of a gas at a pressure of 99.0 kPa is 300mL. If the pressure increases to 188kPa...
A:
Q: question 12 Calculate the initial molarity of a generic amine, R3N (Kb = 7.23x10-6), whose pOH = 2....
A: Here we have to calculate the initial molarity of a generic amine, R3N where we have given that the ...
Q: a. NH2 + H2N CI hexamethylene diamine b. CI CH2 vinyl chloride
A: The reactions given are,
Q: Problem 5.4 = Cl, Identify the chirality centers in the following molecules (yellow-green pale yello...
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in molecules in th...
Q: What is the change in Gibb's Free Energy for a reaction that has an enthalpy change of -270.8 kJ/mol...
A: The change in enthalpy is equal to the energy supplied as heat at constant pressure. The equation ΔG...
Q: Bang-O is known to be an organic pesticide that contains 10.45% Cl. A 0.827 g sample containing the ...
A: Here we are required to find the percentage of pesticide in the sample
Q: 8
A:
Q: g. phosgene + HCI но HO,
A:
Q: How many moles of K are there in a sample of K that contains 1.14x1024 atoms? moles Submit Answer Re...
A: The question is based on mole concept According to Avogadros no. 1 mole of any substance contai...
Q: e. HCOOH + MNO4¯→CO, + Mn2* in acidic solution
A:
Q: Show how the wittig reaction might be used to prepare the following alkene. Draw the alkyl bromide a...
A:
Q: (CH3CO)20 Br2 a.) H2N- CH3 pyridine FeBr3 1. LIAIH4, ether 2. Но b.) NaN3 Br c.) H2/Ni + -NH2
A: Here we are required to predict the product of reaction.
Q: O,N CAty Br febrs
A: The reaction given is,
Q: asing acid strength. them
A:
Q: Magnesium metal reacts with hydrochloric acid to produce hydrogen gas and a solution of magnesium ch...
A: Balanced chemical equation can be defined as the equation in which equal number of atoms of every el...
Q: What the time l submevs ion in day ot Brass alloy (25%2n, 45%. cu) , avea was 10 cm2, that 2.5 Kg ノ ...
A: Given, Brass alloy having 25%zinc and 75%copper Area-10cm2 Weight of NACL displaced by brass-2.5kg ...
Q: Copper can be electroplated at the cathode of an electrolysis cell by the half-reaction: Cu2+(aq)+2...
A:
Q: In each row, pick the compound with the bigger lattice energy. Note: lattice energy is always greate...
A:
Q: Consider the reaction 2 S(g) + 3 O2(g) → 2 SO3(g). Using the standard enthalpies of formation listed...
A: Here we have to determine the average bond energy of S=O . For this we have to determine enthalpy o...
Q: (c) Estimate the wavelength of electrons that have been accelerated from rest through a potential di...
A: Given : Potential different by which electron is accelerated = 75 kV = 75000 V
Q: A 3.40 gram mixture of potassium chlorate KCLO3, and potassium chloride, KCL, was heated until all o...
A: Given, KClO3(s) ➝ KCl(s) + O2(g) Balancing the above equation, 2KClO3(s) ➝ 2KCl(s) + 3O2(g) Pressure...
Q: Q2: OH OH Br Prepare CH;CH,CH-CHCH3 starting from CH;CH,CH-CH2CH3 And any other needed reagents.
A: We have to prepare given diol from alkyl halide.
Q: For the reaction 2 sO2(9) + 02(9) 2 S03(g) AG° = -140.1 k) and AS = -187.9 3/K at 307 K and 1 atm. T...
A:
Q: A buffer solution is 0.100 M in both HC¬H5O2 and LICH5O2 and has a pH of 4.19. Which of the followin...
A: Buffer solution is the solution which resist change in pH on addition of small amount of acid and ba...
Q: Using photoelectron spectroscopy, the ionization energy of the least tightly bound valence electron ...
A: Given, Ionisation energy = 5.40 eV Electronic configuration of Li = 1s22s1 Principal quantum numbe...
Q: Calculate the actual, physiological deltaG for the reaction Phosphocreatine + ADP > creatine + ATP a...
A:
Q: The equilibrium constant, Ke, for the following reaction is 2.90x10-2 at 1.15×10³K. 2S03(g) =2s02(g)...
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical eq...
Q: H;C CH3 -CH=CH-C -CH,CH3 -CH=Ċ- -CH3
A:
Q: For the reaction SbCl (g) SbCl (g) + Cl (g), 5 3 2 G° (SbCl ) = -334.34 kJ/mol f 5 G° (SbCl ) = ...
A: We have find out equilibrium constant.
Q: 2. During Amihan season, the crops in Benguet and other Northern Luzon provinces are prone to damage...
A: Two questions based on properties of liquids that are to be accomplished.
Q: Propose a structure for an amine of formula C,H,N, which liberates a gas when treated with NaNO, and...
A: The NMR spectroscopy is an important tool for the determination of the structure of an organic compo...
Q: d. ClO2→ CIO2 + Cl' in acidic solution
A: We will balance the above reaction by Half reaction method .
Step by step
Solved in 2 steps with 2 images

- Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)Why does the final product has the opposite configuration compared to the reactant? Shouldn’t it form OMs first, then OMs gets substituted by Cl- via Sn2 (the 1st inversion of configuration) then the Cl- gets substituted by OCH3- (the 2nd inversion of configuration? To my understanding 2 inversions = same configurationChemistry please provide the flow of electrons aswell!! thank you! using the starting material (on the left) to determine the sythetic route which will be the most reaosnable and effective to theres none, you have to start witth the begin products to get to finish needed by using the minmium reagents and reactions needed to get to the final product
- What is (are) the elimination product(s) obtained from the following reaction? A)only I B)I and II C) I,II, and ,III D) II, III, and IVCan someone explain the answers to 28, 29, and 30? The answers are B, A, A. I understand lindar catalyst makes cis alkenes and Na/NH3 makes trans alkenes but I dont understand how the second step of Br2, OsO4 influences the stereochemistry between the 2 . Will rate quickly if helpful. Thanks!1.Draw the product, please shows step by step in details 2.Explain if (CH3)2CH in this reaction can attach in ortho positon in Friedel Craft Alkylation?? why or why not???
- Write a detailed mechanism for this reaction that accounts for the net retention of the configuration when Ag+ and low concentration of hydroxide are used.1. Discuss the role of the Aldol condensation reaction in the synthesis below. What specific reaction was used in the synthesis? What is the importance of the aldol reaction in the entire synthetic approach? 2. Show the detailed reaction mechanism involved in their corresponding specific parts in this syntheses.Show the SN2 mechanism in the reaction between (S) 2-bromobutane and iodide ion. Label the product and draw a enery diagram for this reaction.

