Chapter21: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
Section21.SE: Something Extra
Problem 35MP
Related questions
Question
100%
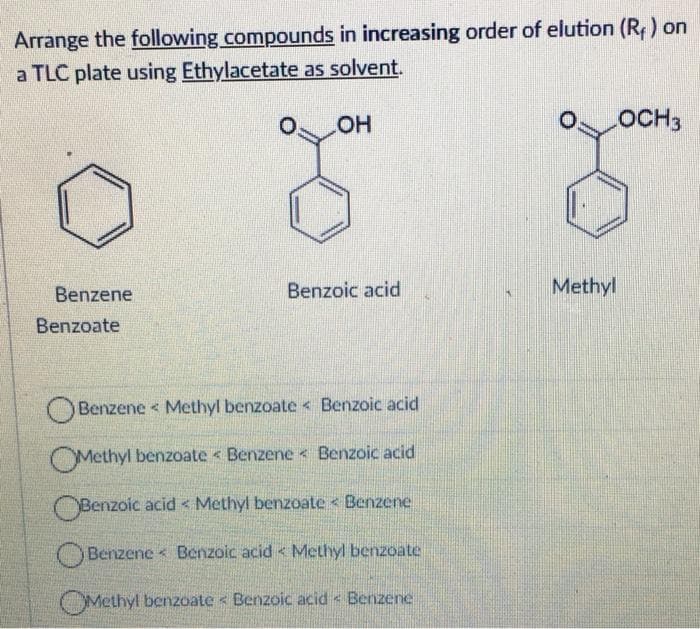
Transcribed Image Text:Arrange the following compounds in increasing order of elution (R) on
a TLC plate using Ethylacetate as solvent.
HO
OCH3
Benzene
Benzoic acid
Methyl
Benzoate
Benzene < Methyl benzoate < Benzoic acid
OMethyl benzoate < Benzene Benzoic acid
OBenzoic acid < Methyl benzoate < Benzene
OBenzene < Benzoic acid < Methyl benzoate
OMethyl benzoate < Benzoic acid < Benzene
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

