Q: What is wrong about the following structures' (hint: there is only one thing wrong in each а. b.
A:
Q: . Find the moleular formula of diazomethane. b. Draw iits lewis structure and resonance forms as…
A: A. moleular formula of diazomethane. b. Lewis structure and resonance forms c. why diazomethane is…
Q: Identify the most stable radical structure. II CH2 CH3 II IV H2C.
A:
Q: Which of the following anions is not stabilized by resonance? (A) (B) (C) (D) OAnion B O Anion A…
A: When double bonds are linked together by the single bond results into the conjugation. similarly…
Q: Use the inscribed polygon method to show the pattern of molecularorbitals in cyclooctatetraene.…
A: When a compound is aromatic, it gains extra stability. Aromaticity was determined by many methods.…
Q: Draw additional resonance structures for each anion.
A:
Q: What is the delocalization energy and π-bond formation energy of (i) the benzene anion, (ii) the…
A: The electronic configuration of benzene is as follows, Then the electronic configuration of benzene…
Q: The Newman projection looking down the indicated bond in the following species is best represented…
A: The given species contains two chiral carbon and the structure of the species is as follows:
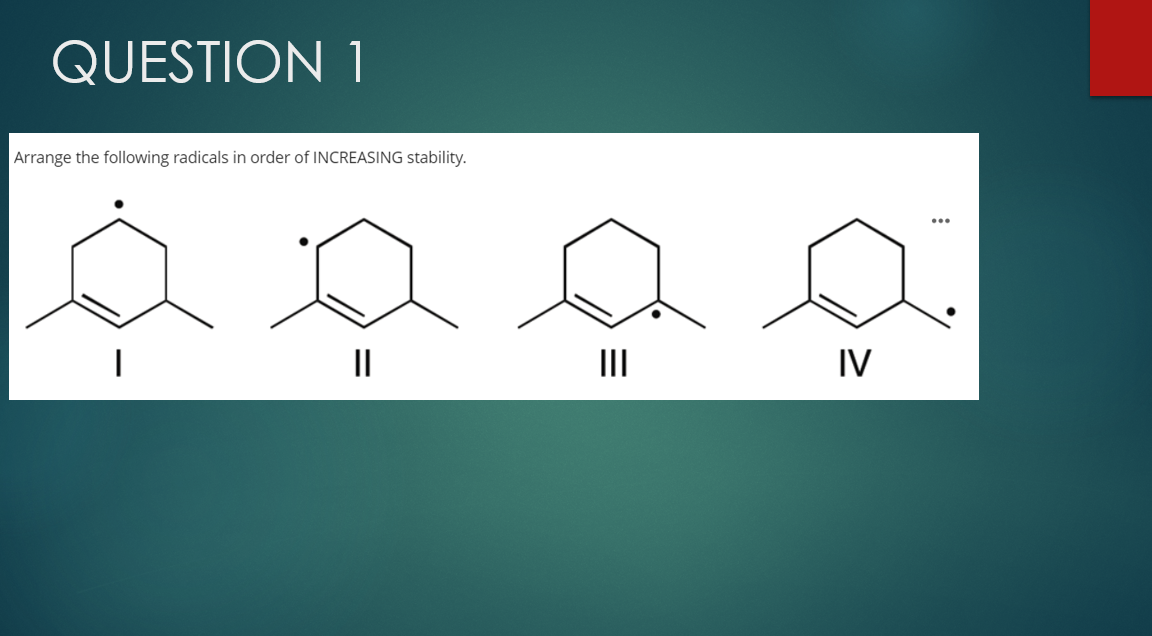
Q: range the following radicals in order of INCREASING stability. II IV IV<1<<II <I1<1<IV IV <Il<1< IIl
A: The given molecules are radical And we know stability of radical depends on the conjugation with…
Q: Arrange the following compounds in order of stability by dragging them into the space below.
A:
Q: List the following radicals in the order of increasing stability.
A: The stability of free radical depends on 2 things. 1) The tendency of free radical to dislocate i.e…
Q: 7. In each set of structures, arrange_the labeled bond s in the order indicaleed Set 1 a Increasing…
A: We have to tell about the bond energy. We know that if a bond having greater s-character, then the…
Q: The most stable MO of 1,3,5-hexatriene and the most stable MO of benzene Which compound is more…
A: Conjugated cycloalkenes has a property of stabilizing the molecule by the delocalization of pi…
Q: Molecule A has 2 resonance forms and molecule B has 3. Numerous experiments have shown that molecule…
A: The contribution of many structures to describe the bonding in any molecule is the phenomenon of…
Q: Arrange the following electron rich species in the increasing order of their stability with an…
A: Electron withdrawing group are those which decreases the electron density and electron releasing…
Q: Which has the greater delocalization energy?
A: Consider the following molecule,
Q: CH Rank the following in order of decreasing importance as a contributing resonance structure to the…
A: Rules :- Rule 1: The most significant resonance contributor has the greatest number of full octets.…
Q: I. II. II.
A:
Q: What are reasons for the low reactivity and high stability of sulfur hexafluoride? (-multiple…
A:
Q: neighboring -CO2H group withdraws electron density inductively and thus
A: We find the reason behind this.
Q: Arrange these structures in decreasing order of reactivity towards bromin ation NH NO 3 O A) 1>3>4>2…
A:
Q: 7. Which radical is most stable? Explain why using structure CH2 CH2
A:
Q: is formed from... Br NH2 N-H (A) (B) (C) (D) (E)
A:
Q: ving free radicals is the MOST stable? C. D.
A:
Q: Classify the radicals into the appropriate categories. Primary,
A: We have to Classify given radicals into primary, secondary, tertiary and allylic radical.
Q: Provide the bond line structure of a compound that is able to undergo Allylic lone pair and allylic…
A: Allylic lone pair: lone pair of an electron adjacent to a double bond. Allylic carbocation: An…
Q: Which is the most likely structure of the following cation after it has rearranged. O A O E OD O B
A: Concept is based on carbocations.
Q: Rank the following radicals in order of decreasing stability (most stable to least stable). II IV A)…
A: Free radicals are odd electrons species having seven electrons in the outermost orbit of the carbon…
Q: Identify the most stable radical structure. H II A) I CH2 CH3 B) II II IV C) II H2C. D) IV
A:
Q: Calculate the energy of isomer B if each 1,3-diaxial interaction costs 3.8 kJ/mol: Br CH -H O 3.8…
A: Presence of any bulky group at axial position will cost two 1,3-diaxial interaction. In the given…
Q: Methotrexate, a drug that inhibits the metabolism of folic acid, is used in the treatment of a…
A: (a) The hybridization of nitrogen atoms marked by star is sp2 hybridized and the other nitrogen…
Q: On the basis of molecular orbital theory and Hückel's rule, which molecules and/or ions should be…
A:
Q: ?Which of the following would you expect to be aromatic H. I II III IV V I and II .a O V b O IV .c O…
A: Aromatic compounds are the compounds that exhibit the following conditions: 1. The molecule should…
Q: Santalbic acid, a fatty acid isolated from the seeds of the sandalwood tree, is an unusual fatty…
A: Given,
Q: Which of the following bonds would have the highest bond dissociation energy? H3C-CI H3C-Br H3C-I A…
A:
Q: Which of the two compounds shown below is more basic? You must rationalize your answer based on…
A: In compound: A, the negative charge lies on an sp hybridized carbon atom B, the negative charge lies…
Q: Rank the following C-C sigma bonds (A, B, C, and D) in order of increasing bond strength. в D
A:
Q: Why does carbon monoxide have a greater bond-dissociation energy than molecular nitrogen
A: In CO molecule: CO show resonating structure in which it consists of 1 triple bond. Also, it has one…
Q: Rank the following bonds in order of decreasing strength (decreasing bond dissociation energies:…
A: Bond dissociation energy (BDE) is the energy needed to make a new molecular fragment by cleavage of…
Q: Which is the most thermodynamically stable state of As? a.H3AsO3 b.H3AsO4 c.AsH3 d.As Which species…
A: Thermodynamic stable means stable due to changes of heat or enegy , it can by with the help of the…
Q: In the molecular orbital model of cyclopentadienyl cation, how many pi-electrons are in bonding…
A:
Q: :0: (а) I or II :ö:0 (b) CH3 II CH3 or (с) CH2 CH2 II or
A: Resonance structure are the structure of a molecule which are obtained by conjugation of π electrons…
Q: CH3 CH3 I. IV. CH3 and and CH3 CH2 I. CH3 CH3 and V. and CH3 CH; II. and
A: A pair of a compound will form isomers if the molecular formula of the molecule is same but differ…
Q: a. Rank molecules A-D (1 = lowest, 4 = highest) in order of increasing boiling point. Briefly…
A: Primary and secondary amines have hydrogen atoms bonded to an nitrogen atom and are therefore…
Q: Arrange the following free radicals in order of increasing stability
A:
Q: CI CH3 CHCHCH3 CH, CHCH CH, • cí CH3 CH3
A: Sigma bonds are the strongest type of covalent chemical bonds . They are formed by head on…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Classify the radicals into the appropriate categories. Primary, Secondary, Tertiary, AllylicBelow is the SN2 reaction between iodocyclohexane and cyanide (CN–). Draw the missing curved arrow notation in the first box to reflect electron movements. In both boxes, add lone pairs of electrons and nonzero formal charges.Ochem reaction scheme question... Provide the bond line structures for the major compounds formed in each step (i, ii, iii, iv, v, heat) of thefollowing reaction scheme (see the attached image)
- All resonance structures with arrow. Major resonance?Provide the bond line structure of a compound that is able to undergo Allylic lone pair and allylic carbocation resonance. Provide all resonance structures and appropriate arrows. Indicate which resonance state is most stable and why.Rank the free radicals shown below in order of increasing stability:

