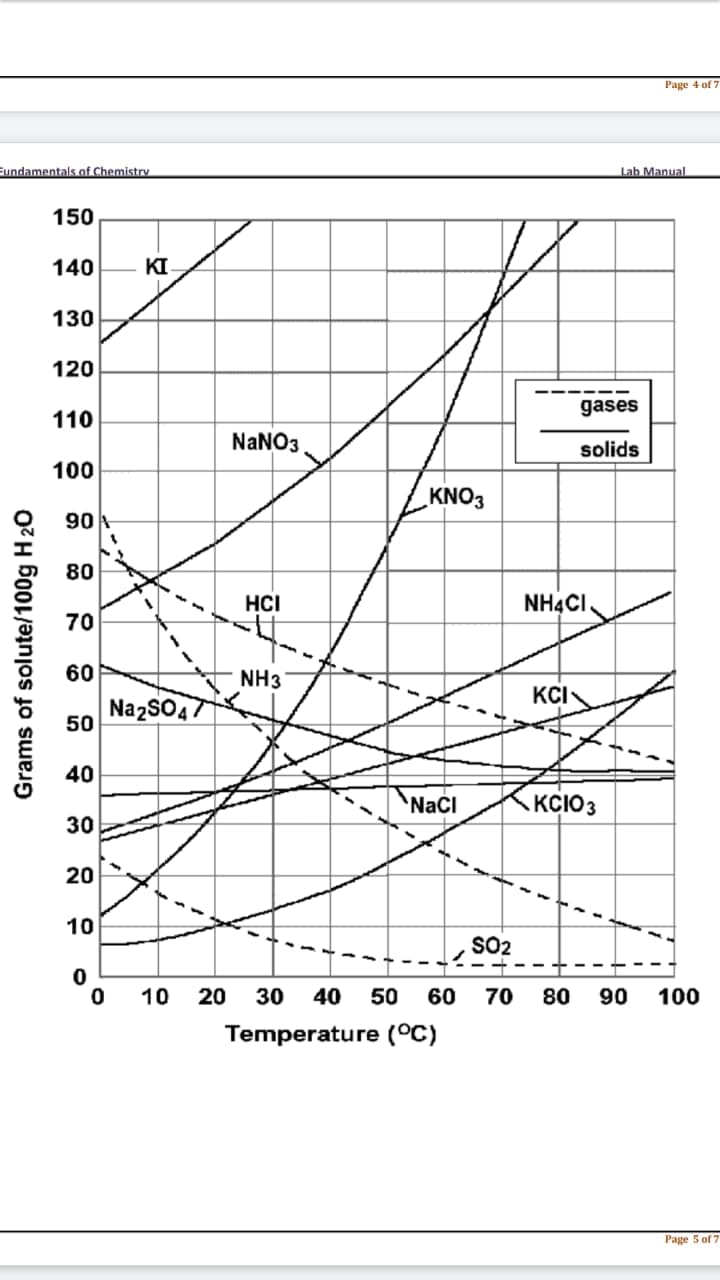
As discussed earlier in solutions involving liquids and solids typically more solute can be dissolved at higher temperatures. Can you find any exceptions on the graph?____________________ 5. A saturated solution of NH4Cl is formed from one hundred grams of water. If the saturated solution is Cooled from 80°C to 40°C, how many grams of precipitate are formed?
As discussed earlier in solutions involving liquids and solids typically more solute can be dissolved at higher temperatures. Can you find any exceptions on the graph?____________________ 5. A saturated solution of NH4Cl is formed from one hundred grams of water. If the saturated solution is Cooled from 80°C to 40°C, how many grams of precipitate are formed?
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter12: Solutions
Section: Chapter Questions
Problem 12.17QE
Related questions
Question
Answer the following questions using the solubility graph:
4. As discussed earlier in solutions involving liquids and solids typically more solute can be dissolved at
higher temperatures. Can you find any exceptions on the graph?____________________
5. A saturated solution of NH4Cl is formed from one hundred grams of water. If the saturated solution is
Cooled from 80°C to 40°C, how many grams of precipitate are formed?
Transcribed Image Text:Page 4 of 7
Eundamentals of Chemistry
Lab Manual
150
140
KI
130
120
gases
110
NANO3
solids
100
KNO3
90
80
HCI
NH4CI.
70
60
NH3
KCI
Na2s04
50
40
NačI
KCIO3
30
20
10
SO2
10
30
40
50
60
70
80
90
100
Temperature (°C)
Page 5 of 7
20
Grams of solute/100g H 20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning