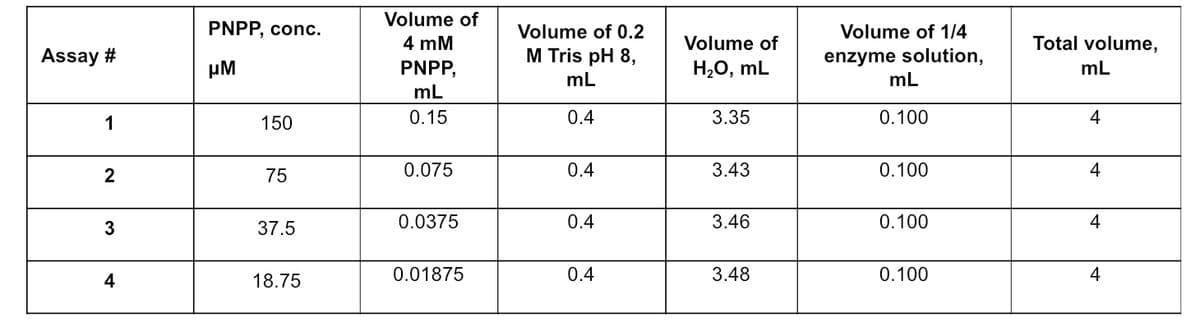
Assay # 1 2 3 4 PNPP, conc. μΜ 150 75 37.5 18.75 Volume of 4 mM PNPP, mL 0.15 0.075 0.0375 0.01875 Volume of 0.2 M Tris pH 8, mL 0.4 0.4 0.4 0.4 Volume of H₂O, mL 3.35 3.43 3.46 3.48 Volume of 1/4 enzyme solution, mL 0.100 0.100 0.100 0.100 Total volume, mL 4 4 4 4
Q: (a) Write the balanced chemical equation that represents the standard heat of formation of Br₂(g) at…
A: The standard enthalpy of formation of a substance is the enthalpy change associated with the…
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI…
A:
Q: What is the electron configurations Mo3+Mo3+
A: The objective of the question is to find the electron configuration of a Molybdenum ion (Mo3+).
Q: Draw the zwitterion of Leucine. CH3 CH₂ CH CH₂ Draw the zwitterion of Leucine.
A: Amino acids contain carboxylic acid and an amine functional group on the same carbon atom. Amino…
Q: Draw the BEST lewis structure (with formal charges) of the indicated chemical species. (Draw all…
A:
Q: What is the electrophile in the Friedel-Crafts acylation reaction below? benzene OCH3CO+ A13+ O 0 Cr…
A: Friedel-Crafts reaction is an electrophilic aromatic substitution reaction. This reaction is used…
Q: atom H K Ca Xe subshell from which electron removed to form +1 cation 1s 11 П 0 subshell to which…
A: Electronic configuration:Electronic configuration can be define as the arrangement of electrons…
Q: For the reaction shown, which of the following answers best describes the product(s) of this…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: 0= H+ H₂O CH 3 Br O-CH3
A:
Q: Magnesium-23 is a positron emitter with a half-life of 11.3039 min. How much of a 1.80-g sample of…
A: Radioactivity is a phenomenon in which nuclei of certain elements undergo spontaneous disintegration…
Q: PROBLEM 18.24 Show how to synthesize the molecule shown here using an aldol reaction. on OH
A: Given,The product molecule:
Q: A chemist adds 130.0 mL of a 7.0 × 10 mmol/L zinc oxalate (ZnC₂04) solution to a reaction flask.…
A:
Q: 5. In question 1, you calculated the enthalpy change for the combustion of propane from combining 3…
A: The objective of the question is to calculate the standard enthalpy change for the combustion of…
Q: Complete the following electron-dot structure for Methylphenidate (C14H19NO2), showing the position…
A:
Q: 1. What is the molarity of 5.30 g of NaCO, dissolved in 400.0 ml solution?
A: Mass of solute (Na2CO3) = 5.30gVolume of solution = 400mL = 400mL x 1/1000 L/mL = 0.4LMolarity =…
Q: Calculate the hydroxide ion concentration ormic acid. (Assume that K¿ (HCOOH) OH-1 of an aqueous…
A:
Q: Starting with the following equation, BCI,(g) + LIAIH (s) → B₂H (g) + LIAICI (s) calculate the moles…
A:
Q: Propose a mechanism for the following transformation Jung l
A: Given is organic reaction.This is Diels-Alder reaction.Diels-Alder reaction is [4+2] cycloaddition…
Q: Draw the major product of the reaction sequence. Omit byproducts. 1. PhCOOOH 2. EtLi, then H3O* 3.…
A: Cyclohexene forms an epoxide when it reacts with peracids. EtLi is a powerful alkylating agent that…
Q: 4. Write in the product of this reaction: I LIAIH
A: LiAlH4 reduces Alkyl cyanide to alkyl amine.
Q: Imagine that you are in chemistry lab and need to make 1.00 L of a solution with a pH of 2.50. You…
A: Molarity of a solution is the measure of its concentration. It is defined as the number of moles of…
Q: A 27.7 mL sample of 0.243 M ethylamine, C₂H5NH₂, is titrated with 0.349 M hydrochloric acid. At the…
A:
Q: Which coordination complexes have a linkage isomer? Select all that apply. O [CO(OH)₂14- O…
A:
Q: From the Lewis structures of the species given, pick all of those in which the central atom obeys…
A:
Q: In EDTA titrations, why does the pH of the solution need to be controlled?
A: Given:In EDTA titrations pH of the solution need to be controlled. We have to explain why pH of the…
Q: Write all the sets of quantum numbers possible for electrons in the n=3 energy level .
A: Quantum numbers are used to represent the position of an electron in an atom. Every electron has a…
Q: Br₂ CH₂Cl₂ H₂ Lindlar Catalyst 1. BD3-Et₂O 2. H₂O₂, NaOH
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Identify, on a labelled diagram, all of the functional groups in the three molecules (A, B and C)…
A: The question is related to functional groups..Functional group: The group of atoms or groups that…
Q: 4. Rhodium has a density of 12.41 g/cm³ and crystallizes with the face-centered cubic unit cell.…
A: Rhodium has a density of 12.41 g/cm3 and crystallizes with the face-centered cubic unit cell.…
Q: Draw the lewis structure with formal charges. If the lewis structure has resonance please show all…
A:
Q: H= -H Br Br
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Draw the Lewis Structures (with formal charges) of each of the species in the indicated pair and use…
A: Order of bond length:Single bond > double bond > triple bondSo, single bonds are longer than…
Q: Your consulting firm wants you to oversee the swimming pool disinfection arm of their business. Your…
A: Answer:Conjugate base of weak acid gets hydrolyzed in water to produce OH- ions, therefore its…
Q: 11. What is the hydrogen ion concentration of a glass of orange juice, with a pH of 2.8? done
A: pH of orange juice = 2.8We need to calculate the hydrogen ion concentration of a glass of orange…
Q: Provide the product(s) for the following reaction: H₂. N₂B
A: Given is organic reaction. this is hydrogenation reaction I.e. reduction reaction. The catalyst used…
Q: Solid potassium cyanide is slowly added to 175 mL of a silver nitrate solution until the…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: Which of these compounds will have a pKa value that is approximately -2? H H O A О в Ос OD Ο Ε e.. :…
A: Acidity is defined as the ability of a molecule to release protons.  If a…
Q: 2) Fill in the missing Reactants, Reagents, b) НО CH₂OH OH OH ОН H2 cat Pd/C H2O Cu²+ Benedict's…
A:
Q: Write an electron-dot structure for chloral hydrate, also known in old detective novels as "knockout…
A:
Q: = to
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Cacodylic acid (dimethylarsinic acid, HASO₂ (CH3)2) is a common buffer used in biology to prepare…
A: The question is based on the concept of the pH of the solution.It is defined as a negative logarithm…
Q: Write the common (not systematic) name of each organic molecule. Hint: your answer should have more…
A:
Q: Consider an atom having BCC structure with atomic weight 167.5 g/mol. The density of the structure…
A:
Q: This figure shows the solubility of gases in water at the same temperature. Use the figure to answer…
A:
Q: A 7.00 L tank at 18.3 °C is filled with 17.0 g of sulfur tetrafluoride gas and 15.2 g of chlorine…
A: According to the question,The mass of the chlorine pentafluoride (ClF5) gas is given by = 15.2 gThe…
Q: 1. Uranium hydride is formed by the reaction of hydrogen gas with solid uranium. The equilibrium…
A: Balanced chemical reaction is given as below:Equilibrium pressure is given as:The equilibrium…
Q: Question 10 of 20 Submit What is Sº for B in the reaction 5 A → 4 Bif AS (rxn) =-218.6 J/mol K? [S°…
A:
Q: N Label C atoms on this structure Carbon Approximate/calculated chemical shift (ppm)
A: NMR spectroscopy is an analytical technique that is used for structural elucidation or…
Q: (2S)-[2-(3,4-Bis-benzyloxy-phenyl)-1-dimethylcarbamoyl-ethyl]-carbamic acid tert-butyl ester BocHN…
A: The given reaction relates to the protection of the phenolic hydroxy group with benzyl chloride.
Q: Write the K₂ expression for an aqueous solution of hypochlorous acid: (Note that either the…
A: The acid equilibrium constant is the ratio of concentration of aqueous product to the concentration…
What is the extinction coefficient for the following absorbances?
1. 3.000
2. 2.022
3.1.2025
4. 0.824
Would the units be in µmoles/mL)^-1 cm^-1
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- You are given three unknown samples stored in screw-cappedcontainers and was told to analyse them. You noted that Container A iscloudy and have some particles that are staying near the bottom of thecontainer. Container B is translucent while Container C is transparent. If thecontents of Container A can be separated by simple decantation,what isthe particle size that BEST describes its composition?i. <1 nmii. 1 nmiii. 1 nm-100 nmiv. >1000 nmMass of aspirin used is .090 g the absorbance for the final solution was .304 nm Slope is 1708 and y-intercept is -0.03397 what is the molarity of the final unknown solution and the molarity of the original solution made from aspirin in the 250.0 ml flaskA. B. and C. already solved (https://www.bartleby.com/questions-and-answers/chemistry-question/a89691d6-2162-4677-b0cc-dc84fcceda08). Please answer E, F and GCircle in pencil in the image on th eirght cide of the diagram is a "2"( Pb(NO3)2 ).
- A chemist obtained the following data for the percent compound Z in triplicates (n=3) of an insecticide preparation: 7.47, 6.98, and 7.27. Calculate the 90% confidence limit for the mean of the data assuming that only information about the precision of the method is the precision for the three data points. 7.24 ± 0.74% 7.24 ± 0.13% 7.24 ± 0.42% 7.24 ± 0.27%what is the mmole of KHP?Chemistry (i) For T = 298K ΔG° = -8.314 x 298 x ln (2.83x10-3) = 14.524 KJ/mol (ii) For T = 308.15K ΔG° = -8.314 x 308.15 x ln (9.619x10-3) = 11.891 KJ/mol (iii) For T = 318.15K ΔG° = -8.314 x 318.15 x ln (3.14x10-2) = 9.147 KJ/mol (iv) For T = 328.15K ΔG° = -8.314 x 328.15 x ln (8.82x10-2) = 6.621 KJ/mol (v) For T = 338.15K ΔG° = -8.314 x 338.15 x ln (2.42x10-1) = 3.990 KJ/mol Based on the measured ΔG° values, is this equilibrium spontaneous at room temperature? Which factor, entropy, or enthalpy, has the greater impact on spontaneity in this case? Explain your answers.
- Please create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72A chemist obtained the following data for the percent compound Z in triplicates (n=3) of an insecticide preparation: 7.47, 6.98, and 7.27. Calculate the 90% confidence limit for the mean of the data assuming that only information about the precision of the method is the precision for the three data points.#9 part E
- What happen to the separation raid (RS) I change the mobile phase from methanol/water 70/30 till methanol/water 80/20. How does polarity affect?Discuss the language demands included in the lesson on mixing substances in a 5th grade classroom by Identify the language function(s) based on the below standard and learning objective. Standard Conduct an investigation to determine whether the mixing of two or more substances results in new substances. Learning Objective Given a variety of substances and safety equipment in a classroom setting, students will be able to conduct an experiment to mix two or more substances and observe the results. They will be able to correctly identify whether a new substance has been formed as a result of the mixing, based on observable changes such as color change, formation of a precipitate, or change in temperature, with at least 80% accuracy.A solution of potassium permanganate of unknown concentration was analyzed against a standard of various concentrations. Refer to the following data set to answer the succeeding questions (MM KMnO4 = 158.034 g/mol). Table 1. Data for Standard Calibration Curve KMnO4 Standard (grams/Liter) Absorbance at 525 nm 0.008 0.10437 0.158 0.20197 0.316 0.45288 0.474 0.68232 0.632 0.89361 Table 2. Data for KMnO4 Solution of Unknown Conc. Sample of Unknown Conc. Absorbance at 525 nm Undiluted KMnO4 0.95423 Diluted KMnO4 (50% dilution) 0.46514 Which of the following linear equation for the standard calibration curve is generated fromTable 1? y= 0.03231x – 0.74923 y= 0.04848x + 1.3179 y= 1.3179x + 0.04845 y= 0.74923x – 0.03231

