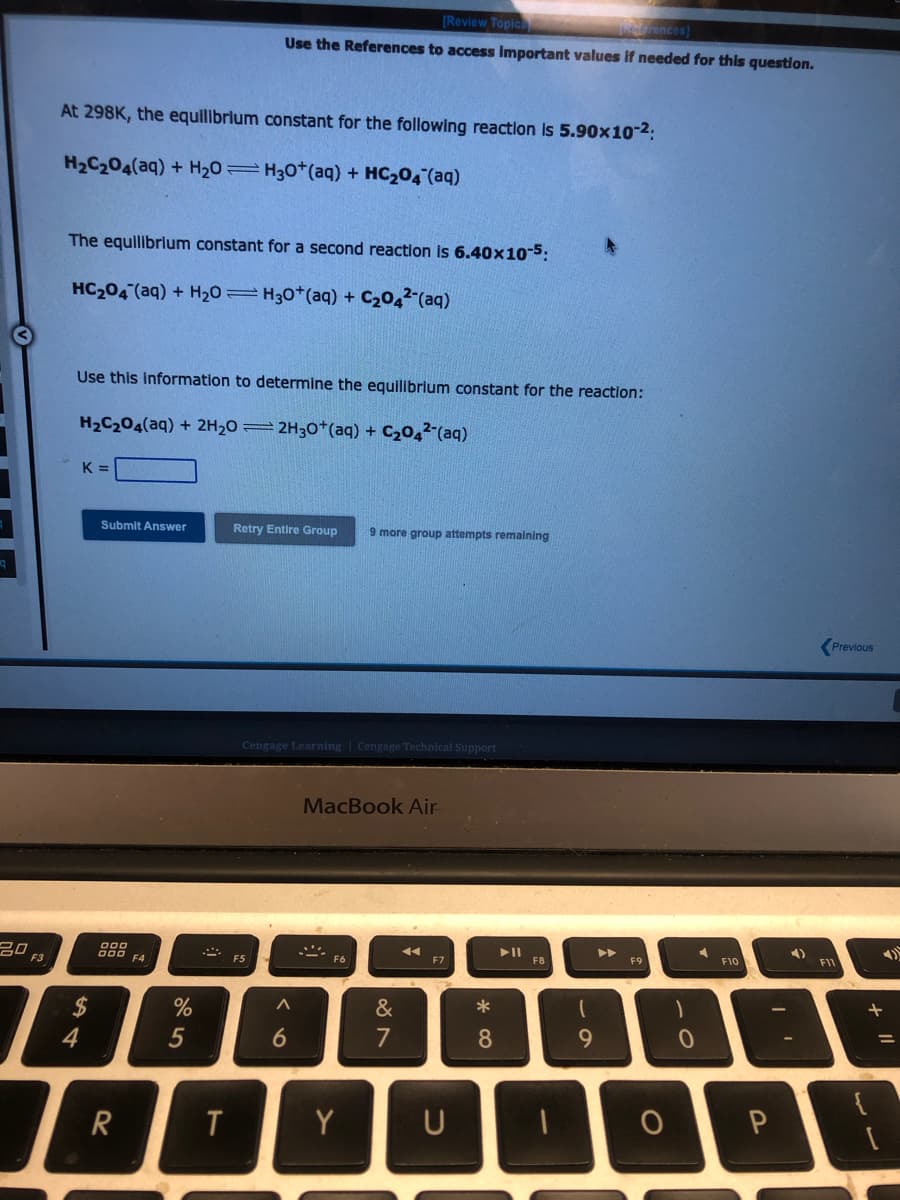
At 298K, the equilibrium constant for the following reaction is 5.90x10-²: H₂C₂04(aq) + H₂O = H3O+ (aq) + HC₂04 (aq) The equilibrium constant for a second reaction is 6.40x10-5: HC₂04 (aq) + H₂0 H30+ (aq) + C₂04² (aq) 4 Use this information to determine the equilibrium constant for the reaction: H₂C₂04(aq) + 2H₂O2H30+ (aq) + C₂04²(aq) K=
At 298K, the equilibrium constant for the following reaction is 5.90x10-²: H₂C₂04(aq) + H₂O = H3O+ (aq) + HC₂04 (aq) The equilibrium constant for a second reaction is 6.40x10-5: HC₂04 (aq) + H₂0 H30+ (aq) + C₂04² (aq) 4 Use this information to determine the equilibrium constant for the reaction: H₂C₂04(aq) + 2H₂O2H30+ (aq) + C₂04²(aq) K=
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter7: Reaction Rates And Chemical Equilibrium
Section: Chapter Questions
Problem 7.32P: 7-32 A particular reaction has an equilibrium constant of 1.13 under one set of conditions and an...
Related questions
Question
Transcribed Image Text:20
F3
At 298K, the equilibrium constant for the following reaction is 5.90x10-²:
H₂C₂04(aq) + H₂0 H30+ (aq) + HC₂04 (aq)
The equilibrium constant for a second reaction is 6.40x10-5:
HC₂04 (aq) + H₂0 H30+ (aq) + C₂04² (aq)
Use this information to determine the equilibrium constant for the reaction:
H₂C₂O4(aq) + 2H₂O2H30+ (aq) + C₂04² (aq)
K=
$
4
Submit Answer
900
R
F4
%
[Review Topics
References)
Use the References to access Important values if needed for this question.
5
T
Retry Entire Group 9 more group attempts remaining
Cengage Learning Cengage Technical Support
F5
A
6
MacBook Air
S
F6
Y
&
7
◄◄
F7
U
*
8
∞
► 11
F8
I
9
►►
O
0
F10
P
Previous
F11
+
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning