At room temperature when HI (aq) is added to potassium bisulfite, KHSO3(s), sulfur dioxide is formed and the reaction vessel becomes cold. HI (aq) + KHSO3(s)→ SO2(g) + H2O(l) + K+(aq) + I-(aq). A.)Once the process has reached equilibrium at room temperature, which of the following must be true? choose one....(ΔH<TΔS, ΔH=TΔS, ΔH>TΔS, or need more info). B.) Once the process has reached equilibrium at room temperature, which of the following must be true? choose one....(ΔSuniv<0, ΔSuniv=0, ΔSuniv>0, or need more info).
At room temperature when HI (aq) is added to potassium bisulfite, KHSO3(s), sulfur dioxide is formed and the reaction vessel becomes cold. HI (aq) + KHSO3(s)→ SO2(g) + H2O(l) + K+(aq) + I-(aq). A.)Once the process has reached equilibrium at room temperature, which of the following must be true? choose one....(ΔH<TΔS, ΔH=TΔS, ΔH>TΔS, or need more info). B.) Once the process has reached equilibrium at room temperature, which of the following must be true? choose one....(ΔSuniv<0, ΔSuniv=0, ΔSuniv>0, or need more info).
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 51QRT: At room temperature, the equilibrium constant Kc for the reaction
2 NO(g) ⇌ N2(g) + O2(g)
is 1.4 ×...
Related questions
Question
At room temperature when HI (aq) is added to potassium bisulfite, KHSO3(s), sulfur dioxide is formed and the reaction vessel becomes cold. HI (aq) + KHSO3(s)→ SO2(g) + H2O(l) + K+(aq) + I-(aq).
A.)Once the process has reached equilibrium at room temperature, which of the following must be true? choose one....(ΔH<TΔS, ΔH=TΔS, ΔH>TΔS, or need more info).
B.) Once the process has reached equilibrium at room temperature, which of the following must be true? choose one....(ΔSuniv<0, ΔSuniv=0, ΔSuniv>0, or need more info).
Expert Solution
Step 1
(A)
The system is at equilibrium means the free energy change of the system is equal to zero. Hence, the relation between enthalpy and entropy change can be obtained as,
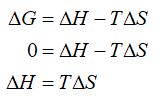
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning
