Average % Chloride: Standard deviation: Show Calculations for Trial 1: a. Mass of Chloride, g b. % Chloride Questions: 1. When the unknown sample was dissolved in 100 mL water, 6 M nitric acid (HNO3) was used to acidify the solution. Why shouldn't any acid such as HCI and H₂SO4 be used in this process? 2. For Trial 1, how many grams of AgNO3 must be added to the solution to completely react with NaCl?
Average % Chloride: Standard deviation: Show Calculations for Trial 1: a. Mass of Chloride, g b. % Chloride Questions: 1. When the unknown sample was dissolved in 100 mL water, 6 M nitric acid (HNO3) was used to acidify the solution. Why shouldn't any acid such as HCI and H₂SO4 be used in this process? 2. For Trial 1, how many grams of AgNO3 must be added to the solution to completely react with NaCl?
Chapter3: Statistical Tests With Excel
Section: Chapter Questions
Problem 5P
Related questions
Question
Please answer the following data with solution.

Transcribed Image Text:Activity No. 8
Determination of Chloride Ion Concentration by Gravimetry
Gravimetric analysis is a method in quantitative analysis where an unknown sample is dissolved
in an appropriate solvent, and the analyte is converted to an insoluble form or precipitation of a known
compound and then weighing the precipitate. In order to do a gravimetric analysis, a cation must be found
that forms an insoluble compound with chloride. Ions such as Ag+, Pb²+, and Hg₂²+ form insoluble chlorides.
Therefore, silver chloride (AgCl) could be used to determine % Chloride (CI), because it is insoluble and it
can be formed pure and is easily filtered.
Ag+ (aq) + Cl(aq) →→ AgCl(s)
One of the most useful applications of gravimetric analysis is the determination of chloride in
natural waters such as seawater and water in rivers and lakes. In this experiment the chlorine content of
an unknown soluble chloride salt is to be determined. The unknown is dissolved in distilled water and
dilute silver nitrate solution is added to precipitate out the chloride as AgCl.
NaCl(aq) + AgNO3(aq) → NaNO3(aq) + AgCl(s)
Note that all of the aqueous species in the above reaction exists as ions in solution:
NaCl(aq) exists as Na+ and Cl
AgNO3 exists as Ag* and NO3
NaNO3(aq) exists as Na+ and NO3
AgCl(s) is a white solid
Procedure Link: https://www.youtube.com/watch?v=dERZhN-01f8
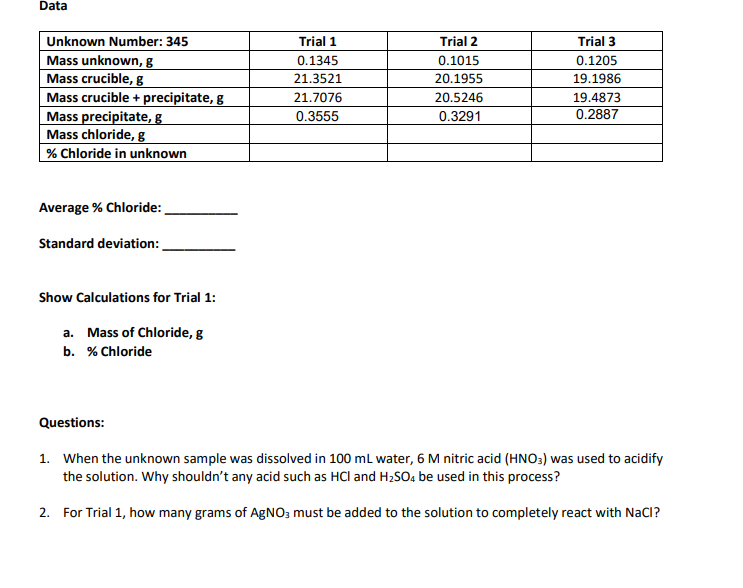
Transcribed Image Text:Data
Unknown Number: 345
Trial 2
Trial 3
Mass unknown, g
0.1015
0.1205
Mass crucible, g
20.1955
19.1986
Mass crucible + precipitate, g
20.5246
19.4873
Mass precipitate, g
0.3291
0.2887
Mass chloride, g
% Chloride in unknown
Average % Chloride:
Standard deviation:
Show Calculations for Trial 1:
a. Mass of Chloride, g
b. % Chloride
Questions:
1. When the unknown sample was dissolved in 100 mL water, 6 M nitric acid (HNO3) was used to acidify
the solution. Why shouldn't any acid such as HCI and H₂SO4 be used in this process?
2. For Trial 1, how many grams of AgNO3 must be added to the solution to completely react with NaCI?
Trial 1
0.1345
21.3521
21.7076
0.3555
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning
