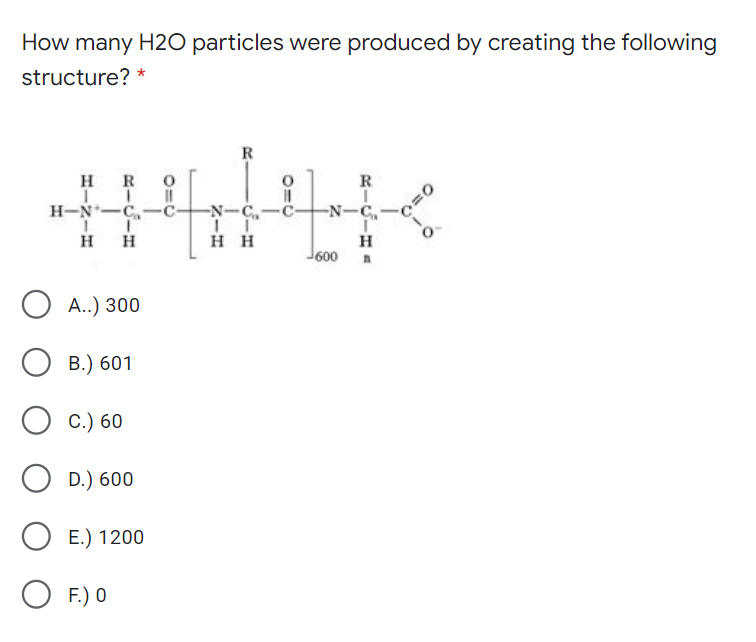
How many H2O particles were produced by creating the following structure? * R H R R H-N-C,-c- -N-C,-C- -N-C,- H H нн H 600
Q: Answer as directed: 1. Which melts at higher temperature? 18:0 or 18:2(Aº. 12) ? Explain. 2. Draw th...
A: Note : Hi ! Thank you for the question. We are authorized to answer one question at a time. Since yo...
Q: You accidentally mixed protease to a solution containing your enzyme, which causes your enzyme to di...
A: Most of the enzymes are proteins. These enzymes may be monomeric or oligomeric. Monomeric enzymes ar...
Q: How many amino acid units do the structure below has? HN. OH OH HO A.) 5 В.) 10 О с.) 9 D.) 8 E.) 7
A: Amini acids are the building blocks of proteins linked through the peptide bond (CO-NH). There are 2...
Q: Drawn below is a schematic of a transmembrane protein. AN Extracellular Cell membrane Cytosolic side...
A: Introduction: A membrane protein is a protein molecule that is connected or linked with the membran...
Q: Ethidium Bromide is a chemical reagent that has been used to detect the presence of nucleic acids. I...
A: Nucleic acids are of two types : DNA and RNA. DNA refers to Deoxyribonucleic acid. It is genetic mat...
Q: 14. Subject this amino acid sequence under (1) trypsin (2) chymotrypsin (3) pepsin and (4) CNBr. Gen...
A: Proteolytic enzymes are used to cleave the larger polypeptide into smaller peptide fragments. They c...
Q: For an individual whose liver is regulated to have gluconeogenesis on and glycolysis off, when energ...
A: Gluconeogenesis is the pathway which synthesize glucose molecules from non-carbohydrate sources such...
Q: What does an A260/A280 ratio of > 1.8 mean?
A: For the DNA sample, an absorbance spectra is obtained in which the maximum absorbance occurs at a pe...
Q: 3. When you think about the primary structure of the mannose-6-P receptor, assuming that it is an in...
A: The internal amino acid sequences in the membrane protein carries the information that determines th...
Q: o Treatment with DNFB gave a mixture of amino acids and DNP-lys o Treatment with trypsin gave an ami...
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer th...
Q: Use the equation of the linear regression line fitted to your standard curve and the migration of yo...
A: Agarose gels are often run to check DNA properties in several molecular biology experiments. The giv...
Q: Starting with pyruvate (CH3COCOO—) as the precursor, the first step of gluconeogenesis is activatio...
A: Gluconeogenesis is a metabolic process that results in the transformation of non-carbohydrate carbo...
Q: What is experiment of Thermodynamics of Small Oligomeric Duplex DNA Denaturation.
A: Double-stranded DNA is the most common kind of DNA molecule found in nature. The strands can be unwo...
Q: Give examples of enzymatic reaction for each type of specificity
A: All biological chemical reaction processes in living creatures are carried out by enzymes. If enzyme...
Q: Calculate the elution volume of blue dextran (blue), myoglobin (red), and Bromocresol purple (purple...
A: Chromatography is separation method of mixture of compounds using column, paper etc, that is based o...
Q: The enzyme phosphofructokinase transfers a phosphate from ATP to the hydroxyl group a C1 of fructos...
A: Substrate specificity - The particular enzyme acts only on it's specific substrate and binds only to...
Q: Calculate the net charge of the molecule at pH 3.
A: Amino acids are organic compounds with two functional group namely carboxyl and amino. Amino acids f...
Q: What would be the expected effect of this change on the enzyme’s catalytic activity?
A: Enzyme activity is the rate of the reaction catalysed by the enzyme expressed. It is measured as mic...
Q: Describe in as much detail as you can, the fluid mosaic model of a cellular membrane.
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: 50 Which best describes the role of FADH2 in aerobic metabolism? O FADH2 is a proton shuttle and tra...
A: Metabolism includes biosynthesis/ reduction (an anabolic process) and oxidation (catabolic p...
Q: The initial pH of a 0.2M arginine solution is 14. a. What is the predominant structure of arginine i...
A: Arginine is a basic amino acid and it contains a guanidino group in its side-chain. Guanidino group ...
Q: How does the summary equation for metabolism relate photosynthesis and cellular respiration? Ph...
A: Carbohydrates are a major forms of energy for both animals and plants. While plants have the ability...
Q: The segment of DNA shown in the figure has restriction sites I and II, which create DNA restriction ...
A: Agarose gel electrophoresis is generally used to separate the DNA molecules. The cathode or negative...
Q: Create a graph illustrating the activity of an enzyme or rate of reaction with the different factors...
A: Rate of reaction is the amount of product formed by the enzyme per unit time . Thisbis expressed in ...
Q: Fibrous proteins characteristically have a hydrophobic interior and a hydrophilic surface. Glycopr...
A: Protein structure is the three dimensional arrengments of polymers specifically polipeptide chain wh...
Q: Which of the following statements concerning structural characteristics of bile acids is CORRECT? * ...
A: Bile acids are a collection of acidic steroid molecular entities with unusual physical, chemical, an...
Q: (a) From the list below, select the amino acid(s) that might by more common in the extracellular dom...
A: Amino acids are the building blocks of proteins which is linked by peptide/amide bond to form polype...
Q: How many mL of 0.254 M NaOH are needed to fully react with 37.50 mL of 0.358 M H2SO4?
A: Concentration of the NaOH = 0.254 M Conc. of H2SO4 solution = 0.358 M or conc. of [H+]=2×0.358 M =0....
Q: Outline the DNA extraction steps?
A: The extraction of DNA is an important initial step in the experiments of recombinant DNA technology.
Q: Compare and contrast the following protein characterization techniques in terms of the principles go...
A: A Physical method for the separation of compounds is called chromatography. In two diff...
Q: What would be the pH level for an enzyme, that prefers acidic conditions.
A: The acidity or basicity of water is determined by its pH. The scale runs from 0 to 14, with 7 denoti...
Q: Aspirin reduces inflammation and prevent platelet aggregation by inhibiting the formation of which o...
A: Aspirin is nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammation. Aspirin ...
Q: The common amino acid residues in B-turis are proline and lysine
A: Proteins are the polymers of amino acids. There are four levels of structural organizations for the ...
Q: Mixtures of amino acids can be analyzed by first separating the mixture into its components through ...
A: Amino acids that take part in protein synthesis are of twenty different types. These twenty amino ac...
Q: 1. Given the mRNA: 5'AUGCAUGACGAUCUCGUCGCG...3' a. Use the genetic code to predict the amino acid se...
A: Introduction: A genetic code is a dictionary that corresponds with the sequence of nucleotides and ...
Q: Two peptides, A and B were tested for activity against Enzyme X, a poison found in the venom of mari...
A: Km and Vmax of an enzyme are calculated from lb plot which is also called as double reciprocal plot....
Q: label all 5 identified regions in the active site, (labels like basic catalytic residue, acidic cata...
A: In the given image tricarboxy-propanol-1 is present at the active site of enzyme and is surrounded b...
Q: 5. Assume that you are trying to do a gel mobility shift experiment using a normal set of Trp genes....
A: The gel mobility shift assay is a biochemistry technique used to detect protein complexes with DNA. ...
Q: 55 Mutations in the gene encoding the glycolytic enzyme triose phosphate isomerase lead to severe me...
A: Introduction: Triose phosphate isomerase deficiency is a severe disorder that is characterized by s...
Q: Given the active site and reaction mechanism below, what is the mechanism of rreversible inhibition ...
A: The mechanism followed by the given example is of Transition state analog Chemical compounds with a ...
Q: How can we use software to identify restriction-enzyme cutting sites in sequenced DNA ?
A: Restriction analysis is the process of identifying restriction mapping sites in DNA sequences by emp...
Q: Why are carbohydrates the body's preferred source of energy? Because they ... * A. are expensive t...
A: Carbohydrates considered as sugar molecules. Along with other proteins and fats, carbohydrates are a...
Q: 1. Predict the membrane orientation of a protein that is synthesized with the following features: (D...
A: The insertion of membrane proteins in their right orientation is decided by multiple sequences that ...
Q: In the third video, glucose, glycine and maltose are tested as samples with the Benedict's reagent. ...
A: Introduction: The benedict is used for the detection of simple carbohydrates. It identifies the redu...
Q: Analysis of a peptide antibiotic purified from a strain of Bacillus brevii resulted in the following...
A: The average molecular weight of 1 amino acid is 110 Da. It is given that the molecular weight of the...
Q: What does a graph of fluorescent relative units (RFUs) vs. time (s) tell about kinetic parameters fo...
A: Fluorescent relative unit (RFU) : It is a unit of measurement employed for DNA Analysis in electrop...
Q: Assessing a remedy for flatulence Certain foods, particularly beans and legumes, contain substance...
A: Carbohydrates, often known as saccharides, are sugar molecules. Carbohydrates are one of three prima...
Q: Give your opinion on whether Gene therapy should not be used in humans. And give some ethical issues...
A: Gene therapy uses the approach of cloning normal genes in the viral vectors. The viral vectors are t...
Q: 2. Draw the structure of the tripeptide Gly-Ala-Tyr H H. N-C-C H N-C-C + N-C-C + H он он CH3 H. 0-H ...
A: Proteins have four levels of structural organization including Primary, secondary, terti...
Q: Give the chemical principles involved in a positive Biuret test, Millon’s Test and Sakaguchi Test. W...
A: The color reactions of the proteins are useful in identifying the amino acids present in them. The c...
Step by step
Solved in 3 steps with 1 images

- How many milliliters of 0.0050 N KOH are required to neutralize 25 mL of 0.0050 N H2SO4? To neutralize 25 mL of 0.0050 M H2SO4?Are Zosyn and pantoprazole compatible in a syringe? If so, at what time and temperature? If not, what reaction occurs?The standard heat of combustion of liquid methyl cyclopentane, C6H12(l),C6H12(l), was measured to be −3937.7 kJ/mol.−3937.7 kJ/mol. What is Δ?̂ ∘f C6H12(l),ΔH^f C6H12(l)∘, the standard heat of formation of liquid methyl cyclopentane?
- If 4.00 L of ethyl alcohol at 18.0 ⁰C is exposed to a temperature of 25.0 ⁰C, what is the resulting volume? (Alcohol: β = 1100x10⁻⁶ / ⁰C) * a. 4.03 L b. 4.31 L c. 0.308 L d. 0.0308 L9.102 O, is reduced when it is converted into O2 . Explain.What mass of nitrogen monoxide would be produced by complete reaction of 17.0 g of ammonia?









