Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter20: Dienes, Conjugated Systems, And Pericyclic Reactions
Section: Chapter Questions
Problem 20.25P
Related questions
Question

Transcribed Image Text:b) What glassware and lab equipment would you need?
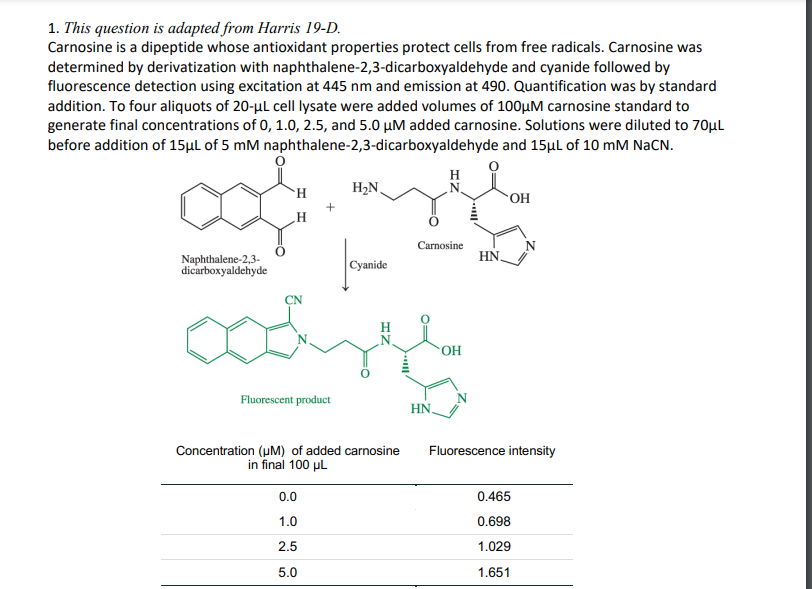
Transcribed Image Text:1. This question is adapted from Harris 19-D.
Carnosine is a dipeptide whose antioxidant properties protect cells from free radicals. Carnosine was
determined by derivatization with naphthalene-2,3-dicarboxyaldehyde and cyanide followed by
fluorescence detection using excitation at 445 nm and emission at 490. Quantification was by standard
addition. To four aliquots of 20-ul cell lysate were added volumes of 100µM carnosine standard to
generate final concentrations of 0, 1.0, 2.5, and 5.0 µM added carnosine. Solutions were diluted to 70µl
before addition of 15µl of 5 mM naphthalene-2,3-dicarboxyaldehyde and 15µl of 10 mM NaCN.
H
H2N.
OH
Carnosine
HN.
Naphthalene-2,3-
dicarboxyaldehyde
Cyanide
OH
Fluorescent product
HN.
Fluorescence intensity
Concentration (uM) of added carnosine
in final 100 µL
0.0
0.465
1.0
0.698
2.5
1.029
5.0
1.651
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning